Amino Acids
Amino Acids
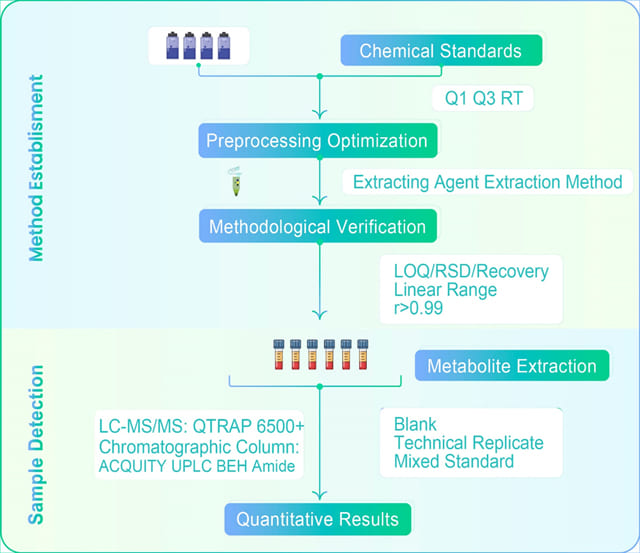
Technology Introduction
Applications of Amino Acid Targeted Metabolomics Service
- Early diagnosis of disease
- Metabolic Disease: Type 2 Diabetes,Obesity
- Neuropathic Disease
- Cardiovascular Disorders(CVD)
- Gastrointestinal Disease
- Nutrition and Health
- Cancer
- Drug development
- Food scientific research
| Amino Acid Compounds | ||
| 2-Aminoethanesulfonic Acid | Sarcosine | γ-Glutamate-Cysteine |
| L-Cystine | L-Pipecolic Acid | Nα-Acetyl-L-glutamine |
| 1,3-Dimethyluric Acid | L-Theanine | N-Acetyl-L-Tyrosine |
| N-Propionylglycine | Ethanolamine | γ-Aminobutyric Acid |
| N-Isovaleroylglycine | 3-N-Methyl-L-Histidine | D-Alanyl-D-Alanine |
| Succinic Acid | Homoserine | Guanidinoethyl Sulfonate |
| 5-Hydroxy-tryptophan | Creatine | Homo-L-arginine |
| 3,7-Dimethyluric Acid | Kinurenine | L-Tryptophyl-L-glutamic acid |
| Glycine | L-Cystathionine | Nicotinuric Acid |
| L-Alanine | 5-Aminovaleric Acid | N-Acetylneuraminic Acid |
| L-Valine | N6-Acetyl-L-Lysine | N,N-Dimethylglycine |
| L-Leucine | Phosphorylethanolamine | 4-Acetamidobutyric Acid |
| L-Methionine | Anserine | L-Carnosine |
| L-Isoleucine | Trans-4-Hydroxy-L-Proline | 6-Aminocaproic Acid |
| L-Proline | D-Homocysteine | 3-Chloro-L-Tyrosine |
| L-Serine | α-Aminoadipic acid | S-(5-Adenosy)-L-Homocysteine |
| L-Tryptophan | L-Ornithine | Kynurenic Acid |
| L-Phenylalanine | L-tyrosine methyl ester | N'-Formylkynurenine |
| L-Tyrosine | 2-Aminobutyric acid | Urea |
| L-Cysteine | (5-L-Glutamyl)-L-Amino Acid | argininosuccinic acid |
| L-Glutamic acid | 3-Iodo-L-Tyrosine | 5-Hydroxylysine |
| L-Aspartate | P-Aminohippuric Acid | O-Phospho-L-Serine |
| L-Asparagine Anhydrous | Glycyl-L-Proline | N-Acetylaspartate |
| L-Glutamine | Trimethylamine N-Oxide | L-Homocystine |
| L-Lysine | 1,3,7-Trimethyluric Acid | 3-Aminoisobutanoic Acid |
| L-Histidine | 3-Hydroxyhippuric Acid | Glutathione Oxidized |
| L-Arginine | N8-Acetylspermidine | L-α-Aspartyl-L-phenylalanine |
| L-Threonine | (S)-β-Aminoisobutyric Acid | N-Glycyl-L-Leucine |
| L-Citrulline | S-Sulfo-L-Cysteine | Creatine Phosphate |
| 5-Hydroxy-Tryptamine | Methionine Sulfoxide | glycylphenylalanine |
| L-Homocitrulline | Nα-Acetyl-L-Arginine | 1-Methylhistidine |
| Beta-Alanine | ||
Abstract
Rearing silkworms (Bombyx mori) using formula feed has revolutionized traditional mulberry feed strategies. However, low silk production efficiencies persist and have caused bottlenecks, hindering the industrial application of formula feed sericulture. Here, we investigated the effects of formula feed amino acid composition on silk yields. We showed that imbalanced amino acids reduced DNA proliferation, decreased Fib-H, Fib-L, and P25 gene expression, and caused mild autophagy in the posterior silk gland, reducing cocoon shell weight and ratio. When compared with mulberry leaves, Gly, Ala, Ser, and Tyr percentages of total amino acids in formula feed were decreased by 5.26%, while Glu and Arg percentages increased by 9.56%. These changes increased uric acid and several amino acids levels in the hemolymph of silkworms on formula feed. Further analyses showed that Gly and Thr (important synthetic Gly sources) increased silk yields, with Gly increasing amino acid conversion efficiencies to silk protein, and reducing urea levels in hemolymph. Also, Gly promoted endomitotic DNA synthesis in silk gland cells via phosphoinositide 3-kinase (PI3K)/Akt/target of rapamycin (TOR) signaling. In this study, we highlighted the important role of Gly in regulating silk yields in silkworms.
Abstract
Extracellular vesicles (EVs) play an important role in human and bovine milk composition. According to excellent published studies, it also exerts various functions in the gut, bone, or immune system. However, the effects of milk-derived EVs on skeletal muscle growth and performance have yet to be fully explored. Firstly, the current study examined the amino acids profile in human milk EVs (HME) and bovine milk EVs (BME) using targeted metabolomics. Secondly, HME and BME were injected in the quadriceps of mice for four weeks (1 time/3 days). Then, related muscle performance, muscle growth markers/pathways, and amino acids profile were detected or measured by grip strength analysis, rotarod performance testing, Jenner-Giemsa/H&E staining, Western blotting, and targeted metabolomics, respectively. Finally, HME and BME were co-cultured with C2C12 cells to detect the above-related indexes and further testify relative phenomena. Our findings mainly demonstrated that HME and BME significantly increase the diameter of C2C12 myotubes. HME treatment demonstrates higher exercise performance and muscle fiber densities than BME treatment. Besides, after KEGG and correlation analyses with biological function after HME and BME treatment, results showed L-Ornithine acts as a"notable marker" after HME treatment to affect mouse skeletal muscle growth or functions. Otherwise, L-Ornithine also significantly positively correlates with the activation of the AKT/mTOR pathway and myogenic regulatory factors (MRFs) and can also be observed in muscle and C2C12 cells after HME treatment. Overall, our study not only provides a novel result for the amino acid composition of HME and BME, but the current study also indicates the advantage of human milk on skeletal muscle growth and performance.
Next-Generation Omics Solutions:
Proteomics & Metabolomics
Ready to get started? Submit your inquiry or contact us at support-global@metwarebio.com.


