The isoelectric point (pI) of an amino acid is the pH at which the molecule carries no net electrical charge. At this point, the positive and negative charges are balanced, and the amino acid exists predominantly in its zwitterionic form. Because charge directly affects solubility, mobility, and molecular interactions, the pI is a key concept in biochemistry, protein chemistry, and separation workflows.
In practice, researchers use amino acid isoelectric points to understand charge behavior, estimate separation conditions, and interpret techniques such as isoelectric focusing and ion-exchange workflows. For neutral amino acids, the pI is often calculated by averaging two pKa values, while acidic or basic amino acids require the two pKa values that flank the zwitterionic form. This guide explains the definition, calculation logic, common pI values, and practical applications of amino acid isoelectric points.
- What Is the Isoelectric Point of an Amino Acid?
- pI Table of Common Amino Acids
- Why the Isoelectric Point Matters
- How to Detect the Isoelectric Point
- How to Calculate the Isoelectric Point
- Factors Affecting Isoelectric Points
- Applications in Industry and Research
1. WHAT ARE AMINO ACIDS?
Amino acids are organic compounds that play critical roles as the building blocks of proteins. Each amino acid molecule consists of a central carbon atom (C), also known as the alpha carbon, bonded to an amino group (NH2), a carboxyl group (COOH), a hydrogen atom, and a distinctive side chain commonly referred to as the R-group. It is this R-group that determines the specific characteristics and chemical behavior of each amino acid.
Understanding the properties of individual amino acids is essential for grasping their behavior in various biochemical processes, including their isoelectric points.
1.1 Structure and Properties
The fundamental structure of amino acids is uniform but the variability of the R-group or side chain leads to diverse chemical properties that influence both the physical and chemical properties of the amino acids. Glycine, the simplest amino acid, is unique due to its amphoteric nature and lack of a side chain, which influences its behavior in solution. These properties impact protein structure and function, as the specific side chains can create complex 3D structures through various interactions like hydrogen bonding, hydrophobic interactions, and ionic bonding.
Amino acids can be classified into four main categories based on the characteristics of their side chains: nonpolar, polar, acidic, and basic. Each class plays a unique role in protein architecture and function. Nonpolar amino acids typically reside in the interior of protein molecules, stabilizing their structure through hydrophobic interactions. Polar amino acids, however, tend to be located on the exterior of proteins where they can interact with the aqueous environment through hydrogen bonds.
1.2 Categories Based on Side Chains
The side chains of amino acids allow for their classification into different groups, each with unique properties that influence protein function:
-
Nonpolar Amino Acids: These amino acids have side chains that are primarily hydrophobic. This group includes alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine, and tryptophan, typically found in the core of proteins, sheltered from water.
-
Polar, Uncharged Amino Acids: This category includes serine, threonine, cysteine, asparagine, and glutamine. Their side chains can form hydrogen bonds, making them hydrophilic and favorably positioned on the exterior of proteins interacting with the surrounding water.
-
Acidic Amino Acids: Aspartate and glutamate fall under this category, each possessing a side chain with a carboxylate group (−COO−) that imparts a negative charge at physiological pH, enhancing water solubility. The presence of an acidic side chain, such as those in aspartate and glutamate, significantly influences the isoelectric point of these amino acids.
-
Basic Amino Acids: Lysine, arginine, and histidine, characterized by their amine functional groups, make this group distinctly positive in charge at physiological pH, which can interact with negatively charged molecules and ions.
The diversity in amino acid side chains and their properties not only dictates the structure and function of proteins but also affects the overall metabolism and biochemistry within organisms. Understanding these properties helps in fields such as medicinal chemistry, where amino acids are utilized to design novel therapeutics.
2. WHAT IS THE ISOELECTRIC POINT of an Amino Acid?
.webp)
Figure 1. Principle of isoelectric focusing. Proteins migrate through a pH gradient and stop at the pH corresponding to their isoelectric point (pI).
The isoelectric point, also known as the pH at which a molecule is electrically neutral or a zwitterion's pH, marks a critical equilibrium in the life of an amino acid. This point occurs when the amino acid's positive and negative charges are in perfect balance, resulting in no overall electrical charge. The balance of these charges is intricately tied to the pKa values of the amino and carboxyl groups that cap the ends of these molecular building blocks.
Amino acids, the fundamental units of proteins, feature an amino group (−NH2) and a carboxyl group (−COOH) at their extremities, behaving as weak bases and acids, respectively. The interaction of these groups with the surrounding pH can lead to different ionic states: at lower pH values, the amino group tends to gain a proton, becoming NH3+, and the carboxyl group loses a proton, becoming COO−, which gives the molecule an overall positive charge. Conversely, At higher pH values, the amino group loses a proton to become NH2, while the carboxyl group remains deprotonated as COO−, giving the amino acid an overall negative charge.
At the isoelectric point, however, the amino acid achieves a state of zen-like neutrality known as a zwitterion, where it is adorned with both positive (NH3+) and negative (COO−) charges, but the overall net charge is zero. This neutrality is crucial for various biochemical processes where proteins need to be isolated or analyzed, as it affects their solubility and movement in an electric field.
3. pI Table of Common Amino Acids
The isoelectric point (pI) of an amino acid is significantly influenced by its chemical structure, particularly the nature of the side chain (R group). This unique side chain dictates the amino acid's overall properties, including its charge at various pH levels, which directly impacts the pI value. The 20 standard amino acids, each with its distinct side chain, have isoelectric points ranging from as low as 2.77 to as high as 10.76, reflecting a wide array of chemical environments.
3.1 Table of Common Amino Acids and Their Isoelectric Points
Each amino acid's isoelectric point is determined by the number and types of charged groups present. The collective properties of many amino acids contribute to the overall behavior of proteins, particularly in techniques like isoelectric focusing. Amino acids with acidic side chains generally exhibit lower isoelectric points due to their higher affinity for protons, while those with basic side chains feature higher isoelectric points.
| Amino Acid | Abbreviation | Isoelectric Point (pI) |
|---|---|---|
| Glycine | Gly | 5.97 |
| Alanine | Ala | 6.11 |
| Arginine | Arg | 10.76 |
| Aspartic Acid | Asp | 2.77 |
| Cysteine | Cys | 5.07 |
| Glutamic Acid | Glu | 3.22 |
| Histidine | His | 7.59 |
| Tyrosine | Tyr | 5.66 |
This table provides a clear, comparative view of the isoelectric points, illustrating how the nature of the side chain influences the overall charge and solubility of the amino acids at different pH levels. By manipulating these properties, scientists and engineers can effectively use amino acids in a variety of biochemical applications.
4. Why the Isoelectric Point Matters?
The isoelectric point helps predict how an amino acid or protein will behave under different pH conditions. At the pI, net charge is zero, solubility is often reduced, and migration in an electric field is minimized. This is why pI is directly relevant to protein precipitation, isoelectric focusing, ion-exchange separation, and many proteomics sample preparation or purification workflows.
4.1 Influence on Solubility
Solubility is strongly influenced by molecular charge. In many aqueous systems, amino acids and proteins tend to be least soluble near their isoelectric point because net charge is minimal and electrostatic repulsion is reduced, which can make aggregation or precipitation more likely. As the pH moves away from the pI, the molecule usually carries a greater net positive or negative charge, which often improves solubility. This behavior is one reason why pI is important in protein purification, crystallization, and separation workflows.
4.2 Behavior in Chromatography and Electrophoresis
The isoelectric point also critically impacts the behavior of amino acids during chromatographic and electrophoretic processes, which separate molecules based on charge and size. The protein isoelectric point is crucial in determining the solubility and charge of proteins during these separation techniques. In techniques like isoelectric focusing, proteins are separated within a pH gradient that mirrors the range of protein isoelectric points. As proteins migrate through this gradient, they halt when the pH matches their isoelectric point because they no longer carry a net electrical charge. This cessation of movement allows for the effective separation of proteins based on their isoelectric points, facilitating detailed analysis and purification.
5. HOW TO DETECT THE ISOELECTRIC POINT?
Detecting the isoelectric point of amino acids and proteins is a fundamental task in biochemistry, essential for applications ranging from drug formulation to protein synthesis and purification. The techniques to determine the pI value vary, depending on the molecule's properties and the required precision.
5.1 Isoelectric Focusing (IEF)
Isoelectric focusing is a powerful and precise technique used to determine the pI of proteins. In this method, proteins are separated based on their isoelectric points within a pH gradient established in a gel. The protein migrates through the gel and stops moving when it reaches the region where the pH equals its isoelectric point; effectively focusing the protein at its pI. This technique not only confirms the isoelectric point but also helps in purifying the protein in its most stable and soluble form.
5.2 Capillary Electrophoresis
Capillary electrophoresis is another highly effective method for determining the pI of amino acids. This approach utilizes a capillary tube filled with a buffer that forms a pH gradient. Similar to IEF, the amino acid or protein migrates until it reaches a point in the gradient where the pH is equal to its pI, at which point it stops moving. This method is particularly useful for analyzing small samples and offers high resolution and quick results.
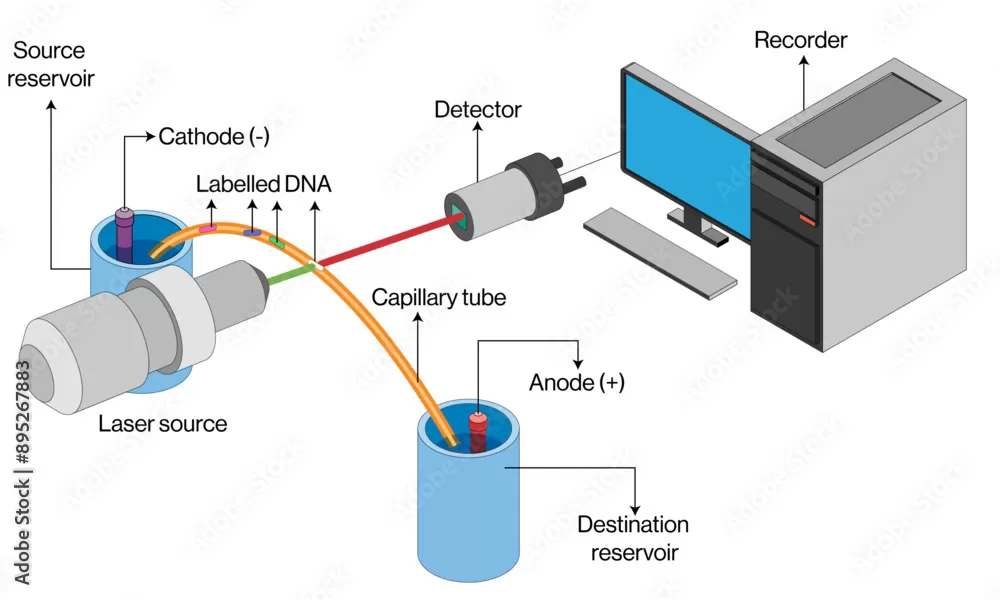
Figure 2. Capillary electrophoresis apparatus for pI determination. The method uses a pH gradient within a capillary tube to separate molecules at their isoelectric points.
5.3 pH Titration
pH titration is a traditional method that involves gradually changing the pH of a solution containing the amino acid while monitoring the charge state of the molecule. The pI is identified at the midpoint of the titration curve, where the molecule exhibits no net electrical charge. This method can be time-consuming and less precise than other techniques but is universally applicable and relatively easy to perform with standard laboratory equipment.
.webp)
Figure 3. 2D separation by capillary isoelectric focusing combined with capillary reversed-phase liquid chromatography — an advanced approach to protein characterization.
6. HOW TO CALCULATE THE ISOELECTRIC POINT
Calculating the isoelectric point (pI) of amino acids is a crucial step in biochemical characterization, impacting applications such as protein purification and enzyme activity studies. Below is a detailed, step-by-step guide that outlines how to determine this critical biochemical parameter.
A simple rule is that the pI is calculated from the two pKa values that surround the zwitterionic form. For neutral amino acids such as glycine, this is usually the average of the alpha-carboxyl and alpha-amino pKa values. For acidic amino acids such as aspartic acid, use the two lower pKa values; for basic amino acids such as lysine, use the two higher pKa values. This logic helps explain why acidic amino acids have lower pI values and basic amino acids have higher pI values.
Example 1: Glycine -> pI = (2.34 + 9.60) / 2 = 5.97
Example 2: Aspartic acid -> pI = (1.88 + 3.65) / 2 = 2.77
Example 3: Lysine -> pI = (8.95 + 10.53) / 2 = 9.74
Step 1: Gather the Necessary Information
Before you begin the calculation, it's essential to collect all necessary data. Primarily, you will need the pKa values of the amino group (−NH2) and the carboxyl group (−COOH). These pKa values represent the acidity and basicity constants associated with their protonation and deprotonation respectively.
Step 2: Identify the Acidic and Basic pKa Values
From the data collected, determine the pKa value for the amino group's acidity (pKa1) and the carboxyl group's basicity (pKa2). The pKa1 is the dissociation constant of the carboxyl group, while pKa2 is for the amino group.
Step 3: Determine the Isoelectric Point
The isoelectric point is calculated using the formula:
pI = (pKa1 + pKa2) / 2
This equation averages the pKa values of the acidic (carboxyl group) and basic (amino group) functional groups, providing the pH at which the amino acid has no net charge (neutral pH).
Step 4: Interpret the Result
The resulting pI value indicates the pH at which the amino acid is electrically neutral. Environmental pH values below the pI will confer a net positive charge to the amino acid, making it more cationic, while pH values above the pI will impart a net negative charge, rendering it more anionic. This knowledge is pivotal in understanding how the amino acid will interact with solvents, substrates, and other molecules.
Step 5: Consider Other Factors
While the pKa values are critical for calculating the pI, other factors such as additional functional groups, molecular modifications, and the ionic strength of the environment can influence the overall charge and behavior of the molecule. These should be considered to adjust the pI calculation accordingly, ensuring accurate predictions and applications in experimental setups.
7. FACTORS AFFECTING ISOELECTRIC POINTS
The isoelectric point (pI) of an amino acid can be influenced by various factors that modify its chemical environment. These factors can be broadly categorized into environmental aspects and chemical modifications. Understanding these influences is crucial for accurately predicting pI values in different contexts, which is essential for applications ranging from pharmaceuticals to biotechnological processes.
7.1 Environmental Factors
Effect of Solvent
The nature of the solvent can significantly affect the isoelectric point of amino acids. Solvents alter the pKa values of the amino and carboxyl groups, primarily through solvation effects. Polar solvents like water increase the dissociation of acidic and basic groups, thus can shift the pI to a more neutral pH, whereas non-polar solvents might decrease this dissociation, resulting in a different pI.
Temperature and Ionic Strength Impact
Temperature fluctuations and changes in ionic strength also play critical roles. Increasing temperature generally increases the dissociation constants (pKa) of amino groups, leading to a higher pI, while increased ionic strength can shield the charged groups of an amino acid, potentially lowering the pI by reducing electrostatic repulsions between like charges.
7.2 Chemical Modifications
Post-Translational Modifications
Post-translational modifications (PTMs) such as phosphorylation, acetylation, and glycosylation can introduce additional charged groups to amino acids or alter existing ones. For instance, phosphorylation adds a phosphate group (PO4³−) that increases the overall negative charge, thereby reducing the pI of the modified amino acid.
Chemical Synthesis and pI Alterations
Chemical synthesis techniques that modify the side chains of amino acids can also impact their isoelectric points. Alterations such as alkylation or esterification can change the nature and charge of side chains, significantly shifting the pI. These changes must be carefully considered when designing peptides and proteins for specific functions and environments.
8. APPLICATIONS IN INDUSTRY AND RESEARCH
The isoelectric point (pI) of amino acids and proteins is more than a theoretical concept; its practical applications permeate various aspects of biotechnology and pharmaceuticals. Understanding the pI can significantly enhance the efficiency of protein purification processes and refine drug design and biomolecule engineering techniques.
8.1 Protein Purification
Protein purification is crucial for a variety of applications in science and industry, including drug development, molecular biology research, and the creation of biotechnology products. The pI of a protein can be exploited to achieve highly efficient purification through techniques such as isoelectric focusing. In this method, proteins are separated in a gel that creates a pH gradient. Proteins stop migrating when they reach the region where the pH matches their isoelectric point, because their net charge becomes zero. This focusing effect enables the separation of proteins with closely related pI values.
8.2 Drug Design and Biomolecule Engineering
The isoelectric point is also integral to drug design and the engineering of biomolecules. In drug design, the pI of a protein can influence its interaction with target receptors, which is critical for the drug's efficacy and stability. Adjusting the pI through molecular modifications can enhance these interactions or improve the solubility and distribution of a drug within the body. For example, modifying the pI of an enzyme can change its substrate specificity, thereby making it more effective for a particular reaction.
In biomolecule engineering, proteins with modified isoelectric points can be designed to form more stable interactions with other biomolecules or to resist changes in environmental conditions such as pH shifts. This is particularly important in creating therapeutic proteins that must remain stable and active under physiological conditions. Manipulating the pI of these proteins can also prevent aggregation when used in therapeutic formulations, which is a major challenge in biologic drug development.
Overall, the practical applications of understanding the isoelectric point in proteins are vast and critical. They not only support the purification and detailed analysis of proteins but also aid in the sophisticated design of drugs and biomolecular therapies. These applications underscore the importance of isoelectric points in advancing both research methodologies and industrial biotechnology.
FAQs
Q1. What is the isoelectric point of an amino acid?
The isoelectric point (pI) of an amino acid is the pH at which the molecule has no net electrical charge. At this pH, the positive and negative charges are balanced, and the amino acid exists mainly in its zwitterionic form.
Q2. How do you calculate the isoelectric point of a neutral amino acid?
For a neutral amino acid, the pI is usually calculated by averaging the pKa values of the alpha-carboxyl group and the alpha-amino group. For example, glycine has pKa values of about 2.34 and 9.60, so its pI is approximately 5.97.
Q3. How is the pI of acidic or basic amino acids calculated?
For acidic or basic amino acids, you do not simply average the lowest and highest pKa values. Instead, you use the two pKa values that surround the zwitterionic form. Acidic amino acids therefore have lower pI values, while basic amino acids have higher pI values.
Q4. Is the isoelectric point the same as pKa?
No. pKa describes the tendency of a specific ionizable group to gain or lose a proton, while pI is the overall pH at which the molecule carries no net charge. A single amino acid can have multiple pKa values but only one isoelectric point.
Q5. Why is solubility often lowest at the isoelectric point?
At the isoelectric point, the molecule has no net charge, so electrostatic repulsion between molecules is reduced. This often makes aggregation or precipitation more likely, which is why pI is important in protein purification and crystallization workflows.
Q6. Why does the isoelectric point matter in proteomics and protein separation?
The isoelectric point affects how molecules move in electric fields and how they interact with buffers, columns, and solvents. This makes pI highly relevant to isoelectric focusing, ion-exchange separation, sample cleanup, protein solubility control, and broader proteomics or protein characterization workflows.
From pI Knowledge to Better Protein Characterization Workflows
Understanding the isoelectric point helps researchers make better decisions in protein separation, purification, and downstream characterization workflows. In practice, pI-related knowledge is often used to support isoelectric focusing, ion-exchange strategies, sample preparation, and broader proteomics analysis.
At MetwareBio, we support researchers with proteomics analysis services, including quantitative proteomics, PTM analysis, and protein characterization workflows. If you are planning a proteomics project and need support with study design, sample preparation, or downstream analysis, our team is ready to help. If you would like to discuss your project, please contact us.
Talk to Our Proteomics TeamRead more
-
Top-Down vs. Bottom-Up Proteomics: Unraveling the Secrets of Protein Analysis
-
Comprehensive Guide to Basic Bioinformatics Analysis in Proteomics
-
Strategies for Selecting Key Interacting Proteins in IP-MS Studies
-
Proteomics sample preparation: Choosing the right extraction methods
References
- Pergande MR, Cologna SM. (2017). Isoelectric Point Separations of Peptides and Proteins. Proteomes, 5(1), 4. https://doi.org/10.3390/proteomes5010004
- Xu X, Liu K, Fan ZH. (2012). Microscale 2D Separation Systems for Proteomic Analysis. Expert Review of Proteomics, 9(2), 135–147. https://pubmed.ncbi.nlm.nih.gov/22462786/




