Matrix spraying is a critical pre-analytical step in spatial metabolomics and a primary determinant of mass spectrometry imaging (MSI) data quality, particularly in MALDI-MSI. By forming a uniform matrix–analyte co-crystal layer on tissue sections, matrix deposition directly influences ionization efficiency and sets the foundation for spatial fidelity, analytical sensitivity, and quantitative robustness. However, this step still presents persistent technical challenges, including coating heterogeneity, inconsistent crystal morphology, and metabolite delocalization, which can compromise signal reproducibility and detection sensitivity in MALDI-MSI–based spatial metabolomics. At the same time, a new wave of matrix deposition strategies—spanning more controllable spraying and sublimation workflows, refined crystallization control, and emerging resolution-oriented approaches—is rapidly improving imaging performance. This blog therefore summarizes the key challenges in matrix spraying, reviews representative new methods that enhance reproducibility and sensitivity, and outlines future trends shaping next-generation MSI workflows. (You may also interested at: maldi spatial metabolomics matrix selection guide)
1. CORE CHALLENGES IN MATRIX SPRAYING FOR SPATIAL METABOLOMICS
The performance of matrix spraying is best evaluated by two outcomes: the consistency of ion generation across the tissue surface and the fidelity with which native molecular distributions are preserved. In real-world spatial metabolomics and MALDI-MSI experiments, three recurring issues most often undermine these goals:
- Uneven matrix coating: When droplet size, spray rate, and deposition flux are not tightly controlled, matrix deposition can become locally over-wet or insufficient, resulting in patchy coverage. This heterogeneity drives pronounced intensity variation within the same section, reduces the robustness of inter-region comparisons, and complicates alignment across technical replicates.
- Uncontrolled crystal size: Variability in crystal growth produces heterogeneous microenvironments at the matrix–analyte interface, and overly large crystals can effectively lower spatial resolution. In addition, they tend to exacerbate ion suppression and obscure low-intensity signals, which is particularly detrimental for low-abundance metabolites.
- Metabolite delocalization: If spraying introduces excessive solvent or drying is delayed, metabolites can undergo lateral diffusion across the tissue surface. This blurs authentic spatial patterns and may generate false-positive or false-negative imaging features, ultimately weakening downstream biological interpretation.
These limitations are especially consequential in clinical and translational settings that demand both sensitivity and spatial precision. In tumor microenvironment studies, for example, patchy matrix deposition can obscure subtle metabolic signatures at invasive margins, while diffusion artifacts can distort the apparent distribution of metabolites across adjacent histological compartments. In neurobiology, even mild delocalization may confound region-specific metabolic interpretation and reduce confidence in pathway-level conclusions drawn from MSI maps. For these reasons, improving matrix spraying is increasingly viewed as a prerequisite for generating robust, publication-quality spatial metabolomics datasets.

Figure 1. Comparison of matrix crystallization between conventional spraying (left) and optimized spraying (right): the conventional condition shows uneven crystal sizes and incomplete coverage, whereas the optimized condition yields finer, more uniform crystals with improved surface completeness.
2. EMERGING MATRIX SPRAYING STRATEGIES FOR BETTER MSI PERFORMANCE
Recent advances in matrix spraying and matrix deposition focus on controlling droplet delivery, stabilizing deposition parameters, and engineering crystal growth to improve both sensitivity and reproducibility in MALDI-MSI. The following four approaches represent distinct—but complementary—routes to addressing crystal uniformity, workflow robustness, and spatial performance.
2.1 Low-Cost Mini-Humidifier Aerosol Coating for Uniform Fine Crystals
A practical and cost-effective strategy uses a mini humidifier to generate solvent aerosol, enabling formation of relatively uniform, small matrix crystals (reported at <10 μm). The core value of this approach lies in lowering the barrier to high-quality matrix coating: it achieves improved deposition uniformity at very low cost (reported < USD 5) and can serve as an accessible alternative to commercial automated sprayers (e.g., ImagePrep) for many laboratories. By tightening crystal-size distribution and improving surface coverage, this method can enhance detection sensitivity and support better spatial resolution in MALDI-MSI, particularly for small molecules and lipids where crystal heterogeneity strongly influences ion yield. Its primary limitation is that performance still depends on practical control of aerosol generation and drying conditions, and the degree of standardization may vary across lab setups compared with fully automated platforms.
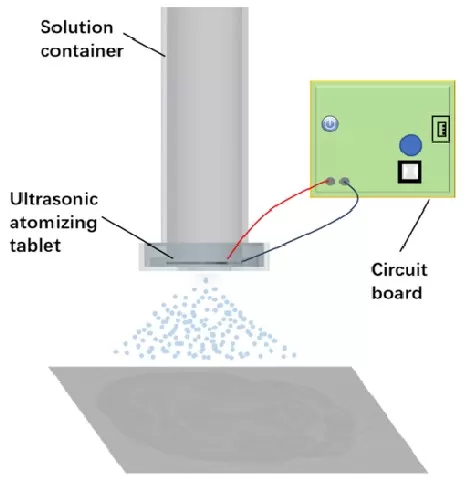
Figure 2. Schematic diagram of the matrix-coating device, made by a mini-humidifier. Image reproduced from Huang et al., 2018, Analytical Chemistry.
2.2 Stainless-Steel Matrix Sublimation Hardware for More Stable Process Control
Hardware redesign is another direction, exemplified by a stainless-steel matrix sublimation device that improves the stability and precision of parameter control during sublimation-based deposition. Relative to conventional glass sublimators, stainless-steel designs can better support reproducible thermal behavior and more consistent operating conditions, translating into improved run-to-run repeatability and shorter preparation time. The key advantage is not only higher workflow efficiency, but also better standardization of the MALDI-MSI matrix deposition process—an important factor when large cohorts, multi-center workflows, or longitudinal studies require consistent performance. The method's emphasis is therefore on boosting reproducibility and operational robustness; however, like other sublimation approaches, success remains sensitive to parameter tuning and may require careful optimization to match different matrices and tissue types.
2.3 Controlled Crystallization Temperature Sublimation for Ultrafine, High-Resolution Coatings
Controlled crystallization temperature (CCT) sublimation extends sublimation strategies by incorporating temperature control—reported to enable ultrafine crystal formation (<0.2 μm) under sub-zero conditions. By promoting highly uniform and extremely small crystals, this approach directly targets the long-standing resolution–sensitivity tradeoffs in MALDI-MSI. Ultrafine and homogeneous coatings can improve imaging sharpness and consistency across tissue types, supporting both high spatial resolution and better signal-to-noise behavior for endogenous and exogenous analytes. The major strength is its ability to deliver high imaging quality and reproducibility through crystal engineering; the practical considerations are that more sophisticated temperature control and tighter process regulation may be required, and the optimal settings can be matrix- and tissue-dependent.
2.4 Gel-Assisted Mass Spectrometry Imaging for Sub-Micrometer Effective Resolution
Gel-assisted mass spectrometry imaging (GAMSI) introduces a conceptually different strategy: rather than solely optimizing deposition, it uses a hydrogel to physically "expand" the sample, effectively improving resolution by 3–6× into the sub-micrometer range without instrument modification. This approach can push spatial metabolomics toward single-cell-level mapping by overcoming pixel-limited constraints of hardware. The key tradeoff is that physical expansion inherently dilutes analyte concentration, which can challenge absolute sensitivity even as apparent spatial detail improves. As a result, GAMSI is powerful for resolution-driven applications, while careful attention is required to balance expansion benefits against the sensitivity costs, particularly for low-abundance metabolites.
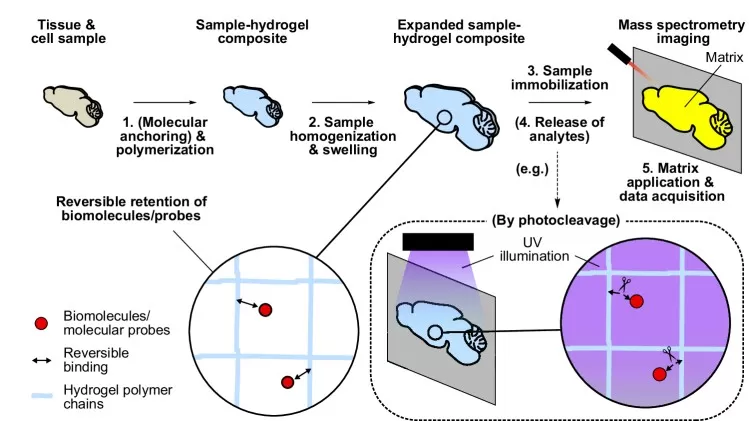
Figure 3. Gel-assisted mass spectrometry imaging (GAMSI). Image reproduced from Chan et al., 2024, Nature Communications, licensed under the Creative Commons Attribution 4.0 International License (CC BY 4.0).
Collectively, these approaches converge on a shared goal: improving MALDI-MSI performance in spatial metabolomics by strengthening matrix deposition as a controllable and reproducible step. Whether through low-cost aerosol deposition, more standardized sublimation hardware, temperature-regulated crystallization, or sample-expansion strategies, each method addresses practical bottlenecks such as crystal heterogeneity, workflow variability, limited sensitivity, and the complexity of high-resolution imaging preparation.
3. FUTURE TRENDS IN MATRIX SPRAYING FOR SPATIAL METABOLOMICS
As spatial metabolomics continues to mature, matrix spraying optimization is expected to evolve from empirical tuning into more standardized, data-driven process engineering. One promising direction is the development of multifunctional spraying systems that improve ionization efficiency through multi-parameter control, including spray direction, pressure, and temperature, with potential integration of physicochemical enhancements such as electric fields or other coupled mechanisms. Such designs aim to further improve coating uniformity while expanding metabolite coverage and strengthening sensitivity for challenging compound classes.
A second direction is deeper automation and intelligent control. By integrating machine learning or AI-assisted decision logic, next-generation systems could infer optimal spraying conditions based on sample characteristics and automatically adjust deposition parameters in real time. Beyond improving reproducibility, these advances may increase throughput and support larger-scale spatial metabolomics studies where consistent matrix deposition is required across many sections and batches.
Finally, innovation in matrix chemistry remains central to improving detection sensitivity. New high-sensitivity matrix materials with broader compatibility, lower analyte loss, and improved ionization behavior may reduce suppression effects and strengthen signal-to-noise for low-abundance metabolites. Nanotechnology-inspired matrices are also being explored as a route to improving both MSI sensitivity and spatial performance, potentially enabling more consistent microcrystal formation and better ion yield under diverse tissue contexts. Together, these developments position matrix spraying not merely as a preparative step, but as a strategic lever for expanding the analytical ceiling of spatial metabolomics across biomedical research, drug development, and other high-impact application areas.
MetwareBio: Your Trusted Partner for Spatial Metabolomics Analysis
MetwareBio provides advanced spatial metabolomics and mass spectrometry imaging (MSI) services for biomedical research, drug discovery, and translational science. With optimized matrix deposition workflows and high-spatial-resolution tissue analysis, we help researchers profile metabolite distributions, map lipid heterogeneity, and investigate the tumor microenvironment with confidence. We also offer Spatial Lipidomics, Neurotransmitter Spatial Profiling and Phytohormone Spatial Profiling to support a wide range of research applications.
If you are interested in spatial metabolomics or MSI-based tissue profiling, please feel free to contact us.
Contact UsReferences
- Wang, X., Han, J., Hardie, D. B., Yang, J., & Borchers, C. H. (2016). The use of matrix coating assisted by an electric field (MCAEF) to enhance mass spectrometric imaging of human prostate cancer biomarkers. Journal of mass spectrometry: JMS, 51(1), 86–95. https://doi.org/10.1002/jms.3728
- Huang, X., Zhan, L., Sun, J., Xue, J., Liu, H., Xiong, C., & Nie, Z. (2018). Utilizing a Mini-Humidifier To Deposit Matrix for MALDI Imaging. Analytical chemistry, 90(14), 8309–8313. https://doi.org/10.1021/acs.analchem.8b01714
- Guo, H., Wu, R., Hao, Q., Qin, L., Chen, L., Hu, H., Feng, J., Wang, L., Liu, R., Chen, D., Wang, A., Wang, Z., Cheng, X., Hao, S., Wang, X., Zhang, H., & Li, G. (2025). Cluster airflow assisted matrix coating enhances high-resolution MALDI imaging of small molecules and lipids in tissues and cells. Chinese Chemical Letters. 112335. https://doi.org/10.1016/j.cclet.2025.112335
- Guo, H., Li, J., Hu, H., Qin, L., Chen, L., Hao, Q., Yang, C., Wu, R., Feng, J., Liu, R., Chen, D., Li, W., Hao, S., & Wang, X. (2026). Alternating Electric Field-Assisted Matrix Nano-Coating for High Spatial Resolution MALDI Imaging. Small methods, 10(3), e00785. https://doi.org/10.1002/smtd.202500785
- Chan, Y. H., Pathmasiri, K. C., Pierre-Jacques, D., Hibbard, M. C., Tao, N., Fischer, J. L., Yang, E., Cologna, S. M., & Gao, R. (2024). Gel-assisted mass spectrometry imaging enables sub-micrometer spatial lipidomics. Nature communications, 15(1), 5036. https://doi.org/10.1038/s41467-024-49384-w
- Zhang, H., Ding, L., Hu, A., Shi, X., Huang, P., Lu, H., Tillberg, P. W., Wang, M. C., & Li, L. (2025). TEMI: tissue-expansion mass-spectrometry imaging. Nature methods, 22(5), 1051–1058. https://doi.org/10.1038/s41592-025-02664-9


