When most people hear the word "cholesterol", they think of cardiovascular risk, clogged arteries, or dietary restrictions. Yet cholesterol is far more than a molecule associated with disease. It is also an indispensable biosynthetic precursor—one that serves as the starting material for every steroid hormone produced in the human body. Steroid hormones regulate a remarkably broad range of physiological functions, including stress adaptation, glucose metabolism, salt and water balance, sexual development, fertility, and pregnancy. Without cholesterol, the body could not synthesize cortisol to coordinate the stress response, aldosterone to maintain blood pressure and electrolyte homeostasis, or sex steroids that drive reproductive maturation and function. In this article, we follow cholesterol from its entry into the steroidogenic pathway to the production of the five major steroid hormone classes.
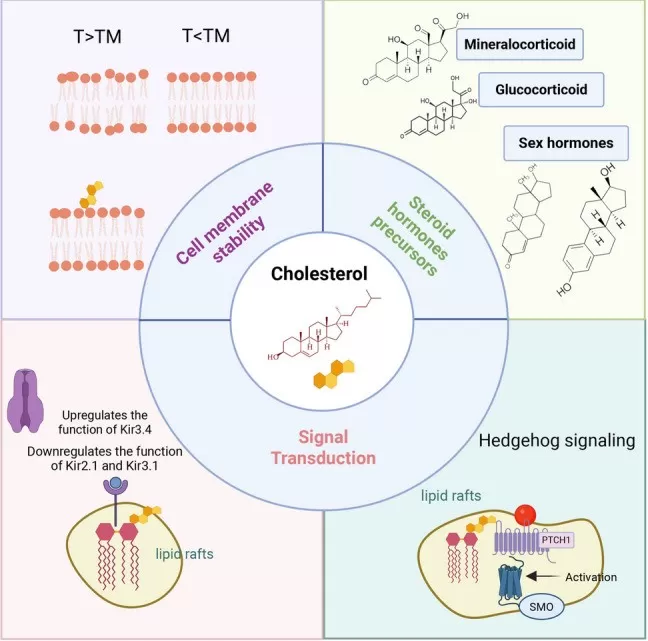
Figure 1. Cholesterol as a multifunctional molecule: beyond membrane structure and cardiovascular health. Image adapted from Cui, D., Yu, X., Guan, Q. et al. (2025), Molecular Biomedicine.
1. THE FIVE MAJOR CLASSES OF STEROID HORMONES
Before examining the pathway itself, it is helpful to understand its major end products. Although all steroid hormones arise from the same precursor, they are grouped into five major classes based on structure, biosynthetic relationships, and physiological function.
| Hormone Class | Primary Representatives | Principal Functions | Major Synthesis Sites |
|---|---|---|---|
| Progestogens | Progesterone | Pregnancy support; precursor for downstream steroid synthesis | Corpus luteum, placenta, adrenal cortex |
| Glucocorticoids | Cortisol | Stress adaptation; glucose metabolism; anti-inflammatory signaling | Adrenal cortex (zona fasciculata) |
| Mineralocorticoids | Aldosterone | Sodium retention; potassium excretion; blood pressure control | Adrenal cortex (zona glomerulosa) |
| Androgens | Testosterone, DHEA | Male sexual differentiation; anabolic effects; libido | Testes, adrenal cortex (zona reticularis), ovaries |
| Estrogens | Estradiol, estrone | Female sexual development; menstrual regulation; bone health | Ovaries, placenta, peripheral tissues |
2. HOW CHOLESTEROL ENTERS THE STEROIDOGENESIS PATHWAY
Steroidogenic cells obtain cholesterol from three principal sources: endogenous de novo synthesis, uptake of circulating lipoprotein-derived cholesterol, and intracellular stores of cholesteryl esters. During acute steroidogenic demand, cholesteryl ester-rich lipid droplets are particularly important because they provide a rapidly mobilizable cholesterol reservoir.
These lipid droplets are not inert storage depots. They are dynamic organelles associated with proteins such as perilipins and vimentin that help coordinate cholesterol mobilization and intracellular trafficking. Evidence from animal models has shown that disruption of these structural and trafficking systems can impair ACTH-stimulated corticosterone production, underscoring the importance of intracellular cholesterol handling in endocrine physiology.
Recent work has also highlighted the importance of the HDL/SCARB1 axis in adrenal steroidogenesis. SCARB1 mediates selective uptake of HDL-derived cholesteryl esters by adrenocortical cells and is particularly important for meeting rapid glucocorticoid demand during stress. Loss of SCARB1 function markedly compromises stress-induced glucocorticoid production, even when LDL is present, indicating that LDL cannot fully substitute for this pathway.
2.1 FROM CHOLESTEROL TO PREGNENOLONE: THE FIRST AND RATE-LIMITING STEP
The first committed step of steroidogenesis is not a chemical conversion in the cytosol, but the delivery of cholesterol to the inner mitochondrial membrane. This transport step is mediated by the steroidogenic acute regulatory protein (StAR), which is widely regarded as the rate-limiting and most tightly regulated step in steroid hormone biosynthesis.
Once cholesterol reaches the inner mitochondrial membrane, it encounters CYP11A1, also known as the cholesterol side-chain cleavage enzyme. CYP11A1 catalyzes a sequence of reactions—20α-hydroxylation, 22-hydroxylation, and cleavage of the bond between carbons 20 and 22—to remove the six-carbon side chain from cholesterol. The product is pregnenolone, a 21-carbon steroid that serves as the universal precursor for all downstream steroid hormones.
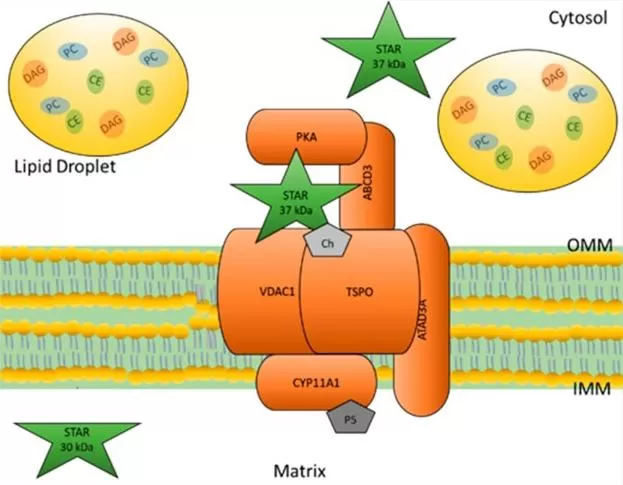
Figure 2. StAR-mediated cholesterol delivery to CYP11A1: the rate-limiting step of steroidogenesis. Image adapted from Galano, M., Venugopal, S., & Papadopoulos, V. (2022), International Journal of Molecular Sciences.
3. THE CORE STEROIDOGENESIS PATHWAYS: Δ5 AND Δ4 ROUTES
From pregnenolone onward, steroidogenesis diverges into two interconnected biosynthetic routes: the Δ5 pathway and the Δ4 pathway. These names refer to the position of the double bond in the steroid backbone. In Δ5 steroids, the double bond lies between carbons 5 and 6, whereas in Δ4 steroids it lies between carbons 4 and 5.
The enzyme 3β-hydroxysteroid dehydrogenase (3β-HSD) is a key branch-point enzyme because it converts Δ5 steroids such as pregnenolone and DHEA into their Δ4 counterparts, including progesterone and androstenedione. CYP17A1—an enzyme with both 17α-hydroxylase and 17,20-lyase activities—serves as another crucial branch point by directing intermediates toward glucocorticoid production or androgen synthesis, depending on tissue context and enzyme activity.
Although the pathway is often drawn as a clean flowchart, steroidogenesis is better understood as a regulated metabolic network. The relative abundance of enzymes, cofactors, and tissue-specific regulatory inputs determines which branch predominates in a given cell type.
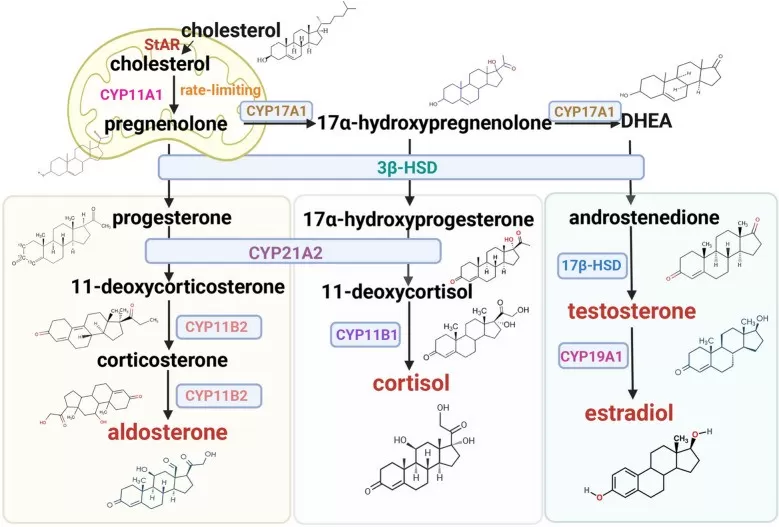
Figure 3. Overview of steroidogenesis: from cholesterol to the major steroid hormones. Image adapted from Cui, D., Yu, X., Guan, Q. et al. (2025), Molecular Biomedicine.
3.1 Progestogens: How Progesterone Is Produced
Progesterone occupies a central position in steroidogenesis. It is generated from pregnenolone by 3β-HSD and functions both as an active hormone and as a precursor for glucocorticoids, mineralocorticoids, and androgens. In reproductive physiology, progesterone prepares the endometrium for implantation, supports maintenance of pregnancy, and contributes to immune tolerance at the maternal-fetal interface.
Key enzyme: 3β-HSD
Core pathway: Pregnenolone → Progesterone
3.2 Glucocorticoids: How Cortisol Is Synthesized
Cortisol is the principal glucocorticoid in humans and is synthesized primarily through the Δ4 pathway. The sequence begins with progesterone, which is converted to 17α-hydroxyprogesterone by CYP17A1, then to 11-deoxycortisol by CYP21A2, and finally to cortisol by CYP11B1.
This pathway is biochemically and physiologically important because cortisol coordinates the systemic response to stress. It promotes gluconeogenesis, modulates immune and inflammatory activity, supports vascular tone, and contributes to circadian regulation of metabolism and arousal.
Core pathway: Progesterone → 17α-hydroxyprogesterone → 11-deoxycortisol → Cortisol
Key enzymes: CYP17A1, CYP21A2, CYP11B1
3.3 Mineralocorticoids: How Aldosterone Is Produced
Aldosterone synthesis takes place primarily in the adrenal zona glomerulosa. In this pathway, progesterone is converted to 11-deoxycorticosterone by CYP21A2, which is then converted to corticosterone and ultimately to aldosterone by CYP11B2, also known as aldosterone synthase.
Aldosterone is essential for sodium retention, potassium excretion, extracellular fluid balance, and blood pressure regulation. Its synthesis is tightly linked to the renin–angiotensin–aldosterone system and serum potassium concentration. Although CYP11B1 and CYP11B2 are highly homologous enzymes, they differ in both expression pattern and regulation. CYP11B1 is expressed primarily in the zona fasciculata and is mainly regulated by ACTH, whereas CYP11B2 is expressed in the zona glomerulosa and is primarily controlled by angiotensin II and potassium.
Core pathway: Progesterone → 11-deoxycorticosterone → Corticosterone → Aldosterone
Key enzymes: CYP21A2, CYP11B2
3.4 Androgens: How DHEA and Testosterone Are Formed
Androgen synthesis can proceed through both the Δ5 and Δ4 routes. In the Δ5 pathway, pregnenolone is converted to 17α-hydroxypregnenolone by CYP17A1 and then to DHEA through the enzyme's 17,20-lyase activity. DHEA is subsequently converted to androstenedione by 3β-HSD and then to testosterone by 17β-HSD. In the Δ4 route, progesterone is first converted to 17α-hydroxyprogesterone and then to androstenedione before testosterone is formed.
These pathways are central to male sexual differentiation, spermatogenesis, and anabolic physiology, but androgens are also biologically important in females, where they contribute to ovarian function, libido, and serving as precursors for estrogen biosynthesis.
Steroidogenesis research has also identified alternative androgen-producing routes, including the so-called backdoor pathway, in which dihydrotestosterone can be generated without testosterone as an obligate intermediate. In addition, C19 11-oxygenated steroids such as 11-hydroxyandrostenedione and 11-ketotestosterone are now recognized as clinically relevant androgens in disorders such as congenital adrenal hyperplasia and polycystic ovary syndrome.
Δ5 pathway: Pregnenolone → 17α-hydroxypregnenolone → DHEA → Androstenedione → Testosterone
Δ4 pathway: Progesterone → 17α-hydroxyprogesterone → Androstenedione → Testosterone
Key enzymes: CYP17A1, 3β-HSD, 17β-HSD
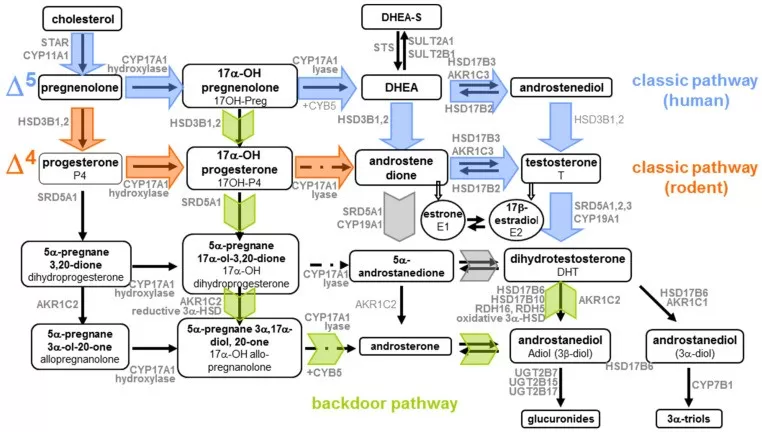
Figure 4. Steroidogenesis pathways: Δ5 and Δ4 routes with the backdoor androgen pathway. Image adapted from Connan-Perrot, S., Léger, T., Lelandais, P. et al. (2021), International Journal of Molecular Sciences.
3.5 Estrogens: How Estradiol and Estrone Are Synthesized
Estrogen biosynthesis depends on aromatase (CYP19A1), the enzyme that converts androgen substrates into estrogens by aromatizing the A ring of the steroid nucleus. Testosterone is converted to estradiol, whereas androstenedione is converted to estrone.
Estrogens play central roles in female sexual development, menstrual cycle regulation, endometrial physiology, bone maintenance, and cardiovascular protection. Aromatase is highly expressed in the ovaries and placenta, but it is also present in peripheral tissues such as adipose tissue, which becomes an increasingly important site of estrogen production after menopause.
Core pathways: Testosterone → Estradiol; Androstenedione → Estrone
Key enzyme: CYP19A1 (aromatase)
4. HOW STEROIDOGENESIS IS REGULATED
Steroidogenesis is tightly regulated at multiple levels, ensuring that hormone output matches physiological demand. Regulation occurs through trophic hormones, intracellular signaling pathways, substrate supply, and tissue-specific expression of steroidogenic enzymes.
4.1 Trophic Hormone Control
ACTH stimulates adrenal steroidogenesis, especially glucocorticoid synthesis, by binding the melanocortin-2 receptor and activating cAMP/PKA signaling. In the gonads, LH and FSH coordinate steroid hormone production through complementary effects on steroidogenic cells and follicular or testicular function. These trophic hormones exert both acute effects—such as rapid stimulation of StAR-dependent cholesterol transport—and chronic effects through transcriptional upregulation of steroidogenic enzymes.
4.2 Enzyme Expression Defines Cell Identity
A steroidogenic cell does not produce a given hormone simply because a precursor is available; it produces that hormone because it expresses the appropriate enzyme repertoire. This principle explains the functional specialization of the adrenal cortex. Zona glomerulosa cells synthesize aldosterone because they express CYP11B2 but not CYP17A1. Zona fasciculata cells generate cortisol because they express CYP17A1 and CYP11B1. The zona reticularis and gonadal steroidogenic cells direct flux toward sex steroids because they support CYP17A1 lyase activity.
4.3 Feedback Regulation
As with most endocrine pathways, steroidogenesis is constrained by negative feedback. Cortisol suppresses hypothalamic CRH and pituitary ACTH release, whereas sex steroids inhibit hypothalamic GnRH and pituitary gonadotropin secretion. These feedback loops stabilize steroid output and prevent inappropriate hormone excess under basal conditions.
5. CLINICAL RELEVANCE: WHEN STEROIDOGENESIS GOES WRONG
Because steroidogenesis depends on a precisely ordered series of enzymatic reactions, disruption at any step can produce major endocrine disease. The resulting phenotype depends on which enzyme is affected, which hormones fall, which precursors accumulate, and whether excess substrate is diverted into alternative pathways.
5.1 Enzyme Deficiencies
21-Hydroxylase deficiency accounts for the overwhelming majority of congenital adrenal hyperplasia (CAH) cases. Deficient cortisol and, in severe cases, aldosterone synthesis lead to ACTH-driven accumulation of precursors, which are then diverted into androgen-producing pathways, causing virilization. Other important defects include 11β-hydroxylase deficiency, which can cause hypertension due to deoxycorticosterone excess; 17α-hydroxylase deficiency, which impairs cortisol and sex steroid production while promoting mineralocorticoid precursor accumulation; 3β-HSD deficiency, which affects both adrenal and gonadal steroidogenesis; and aromatase deficiency, which compromises estrogen synthesis and results in androgen excess.
5.2 Other Endocrine Disorders
Steroidogenic dysregulation is also central to common endocrine diseases. Cushing syndrome results from glucocorticoid excess, whether endogenous or exogenous. Addison disease reflects primary adrenal insufficiency with deficient glucocorticoid and mineralocorticoid output. Apparent mineralocorticoid excess syndrome arises when 11β-hydroxysteroid dehydrogenase type 2 is deficient, allowing cortisol to activate mineralocorticoid receptors inappropriately and drive severe hypertension.
5.3 Therapeutic Implications
A detailed understanding of steroidogenesis has informed multiple therapeutic strategies, including aromatase inhibitors for estrogen-sensitive breast cancer, 5α-reductase inhibitors for benign prostatic hyperplasia, glucocorticoid replacement for adrenal insufficiency, and mineralocorticoid receptor antagonists for hypertension and heart failure.
6. WHY TARGETED STEROIDOMICS MATTERS in Steroidogenesis Research
Steroidogenesis is not a simple linear process ending in a few well-known hormones. Rather, it is a highly interconnected metabolic network in which cholesterol is progressively converted into multiple classes of bioactive steroids through tightly regulated enzymatic steps. Along this pathway, precursor availability, tissue-specific enzyme expression, branch-point control, and feedback regulation all shape the final hormonal output.
This complexity is precisely why pathway-level analysis matters. In both basic research and translational studies, understanding steroid biology often requires more than measuring a single endpoint hormone. A concentration change in cortisol, testosterone, estradiol, or another major steroid may reflect only part of the story. It may not reveal whether the underlying cause is altered precursor supply, enzyme deficiency, compensatory pathway rerouting, or broader metabolic dysregulation.
Traditional hormone testing, while valuable in many contexts, typically focuses on a limited number of individual analytes measured in isolation. This approach can be sufficient for routine assessment, but it may fail to capture the broader biochemical context of steroid hormone biosynthesis.
Targeted steroid metabolomics addresses this limitation by enabling the simultaneous quantification of multiple biologically related steroid metabolites within the same pathway. This broader analytical view helps researchers evaluate precursor-to-product relationships, identify pathway bottlenecks, detect metabolic shunting, and generate more mechanistic insight into endocrine regulation, inflammatory responses, metabolic disorders, and disease-associated signaling.
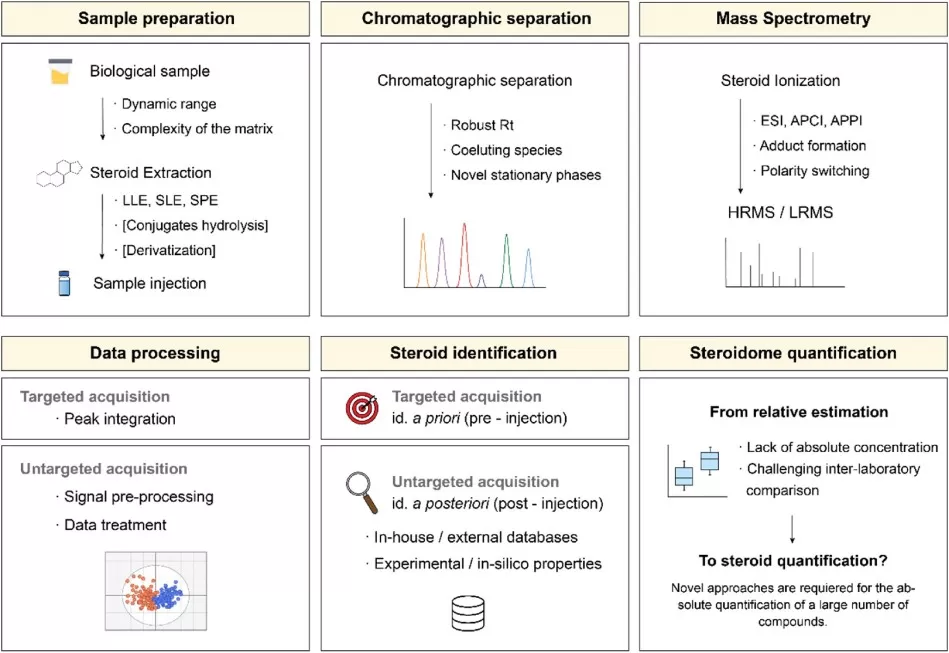
Figure 5. Workflow of steroidome analysis: from sample preparation to quantification. Image adapted from Olesti, E., Boccard, J., Visconti, G. et al. (2021), The Journal of Steroid Biochemistry and Molecular Biology.
MetwareBio: Your Trusted Partner for Steroid Hormone Analysis
To support pathway-oriented research on steroidogenesis, MetwareBio's Steroid Hormones Targeted Metabolomics Service provides a practical and analytically robust solution for the absolute quantification of 40 steroid hormone-related compounds. The panel covers key steroid classes and pathway-associated metabolites, including estrogens, androgens, progestogens, glucocorticoids, mineralocorticoids, cholesterol, and other steroid hormone-related intermediates, enabling a more integrated view of steroid hormone biosynthesis.
Rather than measuring a single end-product hormone alone, this targeted panel allows researchers to profile multiple biologically connected sterol and steroid metabolites within the same analytical framework. Our workflow is supported by 40 external standards and 23 internal standards, enabling robust analytical performance across complex biological matrices.
If you are interested in targeted steroid hormone profiling across major hormone classes or in exploring steroidogenesis through targeted metabolomics, please feel free to contact us for more information.
Contact UsReferences
- Cui, D., Yu, X., Guan, Q., Shen, Y., Liao, J., Liu, Y., & Su, Z. (2025). Cholesterol metabolism: molecular mechanisms, biological functions, diseases, and therapeutic targets. Molecular Biomedicine, 6(1), 72. https://doi.org/10.1186/s43556-025-00321-3
- Kraemer, F. B., Khor, V. K., Shen, W. J., & Azhar, S. (2013). Cholesterol ester droplets and steroidogenesis. Molecular and Cellular Endocrinology, 371(1-2), 15–19. https://doi.org/10.1016/j.mce.2012.10.012
- White, O., Tsujita, M., & Zubiran, R. (2025). The overlooked role of high-density lipoprotein in adrenal stress adaptation: the HDL/SCARB1 axis as a regulator of stress-induced steroidogenesis. Current Opinion in Endocrinology, Diabetes, and Obesity, 32(5), 188–193. https://doi.org/10.1097/MED.0000000000000924
- Bose, H. S., Bose, M., & Whittal, R. M. (2025). Protocol for assessing mitochondrial cholesterol transport and protein molten globule state in steroidogenic and nonsteroidogenic systems. STAR Protocols, 6(3), 103971. https://doi.org/10.1016/j.xpro.2025.103971
- Baronio, F., Ortolano, R., Menabò, S., Cassio, A., Baldazzi, L., Di Natale, V., Tonti, G., Vestrucci, B., & Balsamo, A. (2019). 46,XX DSD due to Androgen Excess in Monogenic Disorders of Steroidogenesis: Genetic, Biochemical, and Clinical Features. International Journal of Molecular Sciences, 20(18), 4605. https://doi.org/10.3390/ijms20184605
- Kater, C. E., Giorgi, R. B., & Costa-Barbosa, F. A. (2022). Classic and current concepts in adrenal steroidogenesis: a reappraisal. Archives of Endocrinology and Metabolism, 66(1), 77–87. https://doi.org/10.20945/2359-3997000000438
- Hanukoglu, I. (1992). Steroidogenic enzymes: structure, function, and role in regulation of steroid hormone biosynthesis. The Journal of Steroid Biochemistry and Molecular Biology, 43(8), 779–804. https://doi.org/10.1016/0960-0760(92)90307-5
- Galano, M., Venugopal, S., & Papadopoulos, V. (2022). Role of StAR and SCP2/SCPx in the Transport of Cholesterol and Other Lipids. International Journal of Molecular Sciences, 23(20), 12115. https://doi.org/10.3390/ijms232012115
- Connan-Perrot, S., Léger, T., Lelandais, P., Desdoits-Lethimonier, C., David, A., Fowler, P. A., & Mazaud-Guittot, S. (2021). Six Decades of Research on Human Fetal Gonadal Steroids. International Journal of Molecular Sciences, 22(13), 6681. https://doi.org/10.3390/ijms22136681
- Olesti, E., Boccard, J., Visconti, G., González-Ruiz, V., & Rudaz, S. (2021). From a single steroid to the steroidome: Trends and analytical challenges. The Journal of Steroid Biochemistry and Molecular Biology, 206, 105797. https://doi.org/10.1016/j.jsbmb.2020.105797


