._1774923737_WNo_387d191.webp)
1. What Is 27-hydroxycholesterol (27-HC) and Why Does It Matter in Cancer?
Cholesterol plays essential roles in human physiology. It is a structural component of cellular membranes, a precursor for steroid hormones, bile acids, and vitamin D, and a critical contributor to brain and reproductive function. However, when cholesterol metabolism becomes dysregulated, it can drive pathological processes, including atherosclerosis, cardiovascular disease, neurodegeneration, and cancer.
Oxysterols are mono-oxygenated cholesterol derivatives generated either enzymatically or through autoxidation. Among them, 27-HC is the most abundant oxysterol in circulation. What makes 27-HC particularly important is its dual role: it contributes to the maintenance of cholesterol homeostasis while also functioning as a signaling mediator capable of influencing tumor biology.
Multiple epidemiological studies have linked elevated cholesterol levels to increased cancer risk, while cholesterol-lowering therapies such as statins have been associated with improved outcomes in certain malignancies. 27-HC offers a plausible mechanistic explanation for part of this association, acting as a molecular intermediary through which cholesterol may exert pro-tumorigenic effects.
2. 27-Hydroxycholesterol Metabolism and Biological Roles in Health and Disease
2.1 Biosynthesis and Catabolism of 27-HC: The CYP27A1-CYP7B1 Axis
27-HC is generated from cholesterol by the mitochondrial enzyme sterol 27-hydroxylase (CYP27A1). This enzyme initiates the acidic, or alternative, pathway of bile acid synthesis and is expressed in a range of tissues, including the liver, lungs, brain, and adipose tissue. The transport of cholesterol into mitochondria, regulated by StAR-related lipid transfer proteins such as StARD1 and StARD3, represents an important rate-limiting step in 27-HC biosynthesis.
Once formed, 27-HC is further metabolized by oxysterol 7α-hydroxylase (CYP7B1), which converts it into 7α,27-dihydroxycholesterol for subsequent processing and eventual bile acid excretion. CYP7B1 therefore serves as a major determinant of intracellular 27-HC abundance. Local 27-HC levels are largely shaped by the balance between CYP27A1-driven synthesis and CYP7B1-mediated catabolism. Accordingly, 27-HC can be viewed as a functional readout of cholesterol turnover and lipid metabolic homeostasis in tissues.
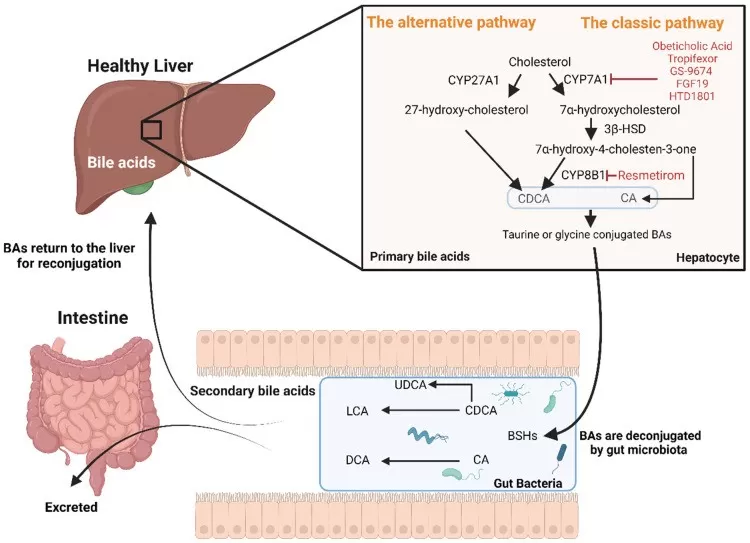
Image reproduced from Bilson, J., Scorletti, E., Swann, J. R. et al., 2024, Biomolecules, licensed under the Creative Commons Attribution License (CC BY 4.0).
2.2 27-HC in Health and Disease: Tissue Distribution, Circulating Levels, and Pathological Dysregulation
Under physiological conditions, 27-HC is one of the most abundant oxysterols in circulation. In healthy individuals, plasma concentrations of free 27-HC generally fall within the range of 0.3 to 0.8 μM, although levels tend to increase with age and in the setting of hypercholesterolemia. Although the liver produces substantial amounts of 27-HC, most hepatic 27-HC is rapidly converted into bile acid intermediates rather than released into the circulation. As a result, a large proportion of circulating 27-HC is thought to arise from extrahepatic tissues.
Pathologically, dysregulation of the CYP27A1-27-HC-CYP7B1 axis has been implicated in multiple diseases, including cancer. In breast and endometrial cancers, for example, CYP27A1 is often upregulated while CYP7B1 is downregulated, leading to intratumoral accumulation of 27-HC. This metabolic imbalance may promote a tumor-supportive microenvironment by amplifying nuclear receptor signaling and altering local cellular behavior.
3. 27-HC Nuclear Receptor Signaling: ER and LXR as Key Targets
The biological effects of 27-HC are mediated primarily through two classes of nuclear receptors: estrogen receptors (ERs) and liver X receptors (LXRs). Through this dual receptor activity, 27-HC acts as a distinctive metabolic messenger that links lipid status to hormone-responsive transcriptional programs.
3.1 27-HC as an Endogenous Estrogen Receptor (ER) Modulator
27-HC was the first endogenous selective estrogen receptor modulator (SERM) to be identified. Unlike synthetic SERMs such as tamoxifen, 27-HC is a naturally occurring cholesterol metabolite that binds estrogen receptors and induces receptor conformations distinct from those triggered by estradiol or pharmacologic ligands.
Its effects on ER signaling are highly context-dependent. In some tissues, 27-HC behaves as an ER agonist, whereas in others it can exert antagonistic effects. In breast cancer cells, however, 27-HC predominantly displays estrogen-like activity, promoting ER-driven transcription and stimulating cell proliferation. This helps explain why elevated 27-HC is especially relevant in hormone-related malignancies.
3.2 27-HC as a Liver X Receptor Ligand
27-HC is also a potent ligand for liver X receptors, LXRα and LXRβ, which function as intracellular cholesterol sensors. Upon activation by 27-HC, LXRs induce the expression of cholesterol transporters such as ABCA1 and ABCG1, thereby promoting cholesterol efflux and contributing to the maintenance of lipid homeostasis. Beyond promoting cholesterol efflux, LXR activation by 27-HC extends to broader metabolic and transcriptional programs that shape the lipid environment of the cell.
In cancer, however, LXR activation can have more complex consequences. LXR-regulated programs influence not only lipid metabolism but also inflammation and immune cell behavior, all of which can be hijacked by tumor cells to support survival, growth, and metastatic progression.
Nuclear receptors targeted by 27-hydroxycholesterol (27-HC) and their roles in cancer:
| Nuclear receptor | 27-HC action | Main effect | Cancer relevance |
|---|---|---|---|
| ERα | SERM (agonist in breast) | Stimulates proliferation | Drives ER+ breast cancer growth |
| LXRα/β | Agonist | Promotes cholesterol efflux, regulates lipid metabolism | Context-dependent: may enhance EMT, invasion, immune evasion |
3.3 A Molecular Bridge Between Metabolism and Oncogenic Signaling
Because 27-HC can signal through both ER and LXR, it serves as a molecular bridge between metabolic state and transcriptional regulation. Through LXR, it conveys information about intracellular cholesterol burden; through ER, it modulates hormone-responsive gene expression. This dual functionality enables cholesterol metabolism to directly influence cancer-relevant signaling pathways.
4. 27-Hydroxycholesterol and Cancer Progression
4.1 27-HC in Breast Cancer: The Strongest Evidence for a Pro-Tumor Role
Among all tumor types studied to date, the role of 27-HC is best established in breast cancer. Preclinical models have shown that dietary cholesterol can promote tumor growth through 27-HC. Once produced, 27-HC stimulates the proliferation, invasion, and metastatic potential of ER-positive breast cancer cells.
Mechanistically, 27-HC acts directly on tumor cells via ERα to promote growth. It also exerts important immunomodulatory effects, influencing myeloid cells, impairing T-cell function, and weakening anti-tumor immune surveillance. In this context, 27-HC can be viewed not only as a signaling metabolite but also as a manifestation of lipid metabolic reprogramming, through which altered cholesterol handling may be linked to proliferation, invasion, and immune evasion in breast cancer. Together, these effects help create a tumor microenvironment that is more permissive to disease progression.
Clinical and translational studies generally support a detrimental role for 27-HC in breast cancer, especially in postmenopausal patients and in more advanced disease. Elevated CYP27A1 expression has been associated with higher intratumoral 27-HC levels and more aggressive tumor behavior. That said, the relationship is not entirely straightforward. Some studies suggest that high CYP27A1 expression may correlate with longer survival in certain subsets of ER-positive breast cancer, particularly in younger women. These findings underscore the complexity and context dependence of 27-HC biology.
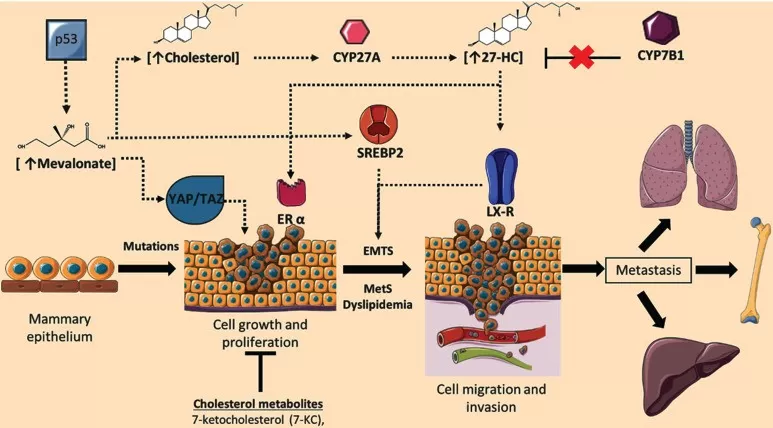
Image reproduced from González-Ortiz, A., Galindo-Hernández, O., Hernández-Acevedo, G. N. et al., 2021, Journal of Cancer, licensed under the Creative Commons Attribution License (CC BY 4.0).
4.2 27-HC in Prostate Cancer: A Tissue-Specific and Emerging Paradox
Prostate cancer presents a striking contrast. While some early preclinical work suggested that 27-HC may support prostate tumor progression, findings from human specimens increasingly suggest a different picture.
CYP27A1 expression is often lost as prostate cancer advances, and restoration of the CYP27A1/27-HC axis has been shown to suppress cancer cell growth. More recent mechanistic work indicates that 27-HC may induce a state of "BRCAness" in prostate cancer cells—a functional impairment of DNA damage repair pathways. This can lead to increased DNA damage and may create therapeutic vulnerabilities, including enhanced sensitivity to PARP inhibition.
The divergent roles of 27-HC in breast versus prostate cancer highlight the critical importance of tissue context. Its effects are not uniformly tumor-promoting. Rather, they appear to depend on the hormonal environment, receptor expression profile, and molecular landscape of a given tumor type.
4.3 27-HC in Other Cancers: Emerging Evidence Beyond Breast and Prostate Cancer
Beyond breast and prostate cancer, the role of 27-HC remains an active area of investigation. In colorectal cancer, advanced-stage tumors have been reported to exhibit increased 27-HC production, potentially supporting tumor cell survival. Endometrial cancer appears to resemble breast cancer in that CYP27A1 is often upregulated and CYP7B1 downregulated, favoring 27-HC accumulation. In lung cancer, thyroid cancer, and glioblastoma, preliminary findings also suggest pro-tumorigenic roles for 27-HC, although the evidence base remains comparatively limited.
5. Clinical Relevance of 27-HC in Cancer Research and Therapy
5.1 27-HC as a Potential Biomarker
Because of its association with tumor progression and metabolic dysregulation, 27-HC has attracted interest as a biomarker for cancer risk stratification and treatment response. Circulating 27-HC levels broadly reflect cholesterol status and may serve as an indicator of cholesterol-related cancer risk. In addition, intratumoral 27-HC abundance, or the expression pattern of CYP27A1 and CYP7B1, may hold prognostic value and potentially inform therapeutic decision-making.
At the same time, the context-dependent biology of 27-HC complicates biomarker development. High CYP27A1 expression may signify adverse biology in breast cancer yet a more favorable phenotype in prostate cancer. Any clinical use of 27-HC-related biomarkers will therefore require careful interpretation within a tumor-specific context.
5.2 Therapeutic Strategies Targeting 27-HC Signaling
Several therapeutic strategies are being explored to attenuate the cancer-promoting effects of 27-HC:
- Inhibiting synthesis: Direct inhibition of CYP27A1 could reduce 27-HC production. Small-molecule CYP27A1 inhibitors are being investigated as potential anti-cancer agents.
- Enhancing degradation: Upregulating CYP7B1 expression or activity may accelerate 27-HC clearance and reduce its local accumulation within tumors.
- Statin therapy: By limiting cholesterol availability, statins can lower 27-HC levels. Clinical studies are evaluating whether this translates into improved cancer outcomes, particularly in breast cancer, although current evidence suggests that systemic lipid lowering does not always produce a direct anti-proliferative effect within tumors.
- Combination approaches: Because 27-HC contributes to multiple mechanisms of treatment resistance—including increased drug efflux, epithelial-mesenchymal transition (EMT), and metabolic reprogramming—combining 27-HC-targeted interventions with standard therapies may be a promising strategy for overcoming resistance.
5.3 27-HC and Drug Resistance
Accumulating evidence suggests that 27-HC may contribute to multidrug resistance (MDR) through several complementary mechanisms. These include promotion of drug efflux, enhancement of EMT, suppression of apoptosis, and rewiring of cancer cell metabolism. A better mechanistic understanding of these processes may reveal new opportunities to restore treatment sensitivity by targeting 27-HC-dependent pathways.
6. Conclusion and Future Perspectives: 27-HC as a Bridge Between Lipid Metabolism and Cancer
27-Hydroxycholesterol sits at a critical intersection of lipid metabolism and cancer biology. As the most abundant oxysterol in human circulation, it functions as both an endogenous SERM and an LXR ligand, linking cholesterol homeostasis to nuclear receptor signaling and tumor-associated processes.
Its role in cancer is highly context-dependent. In breast cancer, 27-HC largely acts as a tumor-promoting metabolite through ER activation, immune modulation, and metastatic reprogramming. In prostate cancer, by contrast, emerging evidence suggests that 27-HC may exert tumor-suppressive effects in certain settings, particularly through mechanisms related to DNA damage and repair. These tissue-specific differences underscore that 27-HC should not be interpreted through a single mechanistic framework.
Looking ahead, several questions remain central to the field. First, the context-dependent actions of 27-HC across different tumor types need to be defined more precisely, particularly to explain why its biological effects differ so markedly between hormone-related cancers. Second, the therapeutic potential of targeting 27-HC metabolism and signaling—including CYP27A1 inhibition, CYP7B1 modulation, and receptor-directed strategies—requires further validation in both preclinical and clinical settings. Third, there is growing interest in combining 27-HC-targeted approaches with endocrine therapy, chemotherapy, immunotherapy, or PARP inhibitors to improve treatment response and overcome resistance. Finally, future studies should clarify whether circulating 27-HC levels, intratumoral metabolite abundance, or CYP27A1/CYP7B1 expression patterns can serve as clinically actionable biomarkers.
In summary, 27-HC is more than a cholesterol-derived metabolite; it is a functional mediator that connects altered lipid metabolism with cancer-related signaling. As research continues to refine our understanding of its diverse and context-specific roles, 27-HC may emerge as both a valuable biomarker and a promising therapeutic target in cancer.
MetwareBio: Supporting Cancer Metabolism Research with Targeted Metabolomics
As discussed in this article, 27-hydroxycholesterol (27-HC) is an important signaling metabolite at the intersection of cholesterol metabolism, nuclear receptor activity, and tumor biology. Because oxysterols are often present at low abundance and are analytically challenging, studies involving 27-HC and related pathways require careful sample handling and reliable LC–MS/MS-based workflows.
MetwareBio supports pathway-focused research in cancer metabolism, lipid metabolism, and hormone-related biology with targeted metabolomics solutions tailored to specific study goals and sample types. Our team can help evaluate suitable analytical strategies for metabolite profiling in translational and mechanism-oriented research.
If you are planning a study related to cholesterol-derived metabolites, cancer metabolism, or pathway-oriented metabolomics analysis, please contact us to discuss the most appropriate solution for your project.
Contact UsReferences
- Hou, Y., Fu, Z., Wang, C., Kucharzewska, P., Guo, Y., & Zhang, S. (2025). 27-Hydroxycholesterol in cancer development and drug resistance. Journal of enzyme inhibition and medicinal chemistry, 40(1), 2507670. https://doi.org/10.1080/14756366.2025.2507670
- Ma, L., Cho, W., & Nelson, E. R. (2022). Our evolving understanding of how 27-hydroxycholesterol influences cancer. Biochemical pharmacology, 196, 114621. https://doi.org/10.1016/j.bcp.2021.114621
- Kakiyama, G., Marques, D., Takei, H., Nittono, H., Erickson, S., Fuchs, M., Rodriguez-Agudo, D., Gil, G., Hylemon, P. B., Zhou, H., Bajaj, J. S., & Pandak, W. M. (2019). Mitochondrial oxysterol biosynthetic pathway gives evidence for CYP7B1 as controller of regulatory oxysterols. The Journal of steroid biochemistry and molecular biology, 189, 36–47. https://doi.org/10.1016/j.jsbmb.2019.01.011
- Umetani M. (2016). Re-adopting classical nuclear receptors by cholesterol metabolites. The Journal of steroid biochemistry and molecular biology, 157, 20–26. https://doi.org/10.1016/j.jsbmb.2015.11.002
- Biasi, F., Leoni, V., Gamba, P., Sassi, K., Lizard, G., & Poli, G. (2022). Role of 27-hydroxycholesterol and its metabolism in cancer progression: Human studies. Biochemical pharmacology, 196, 114618. https://doi.org/10.1016/j.bcp.2021.114618
- Kimbung, S., Chang, C. Y., Bendahl, P. O., Dubois, L., Thompson, J. W., McDonnell, D. P., & Borgquist, S. (2017). Impact of 27-hydroxylase (CYP27A1) and 27-hydroxycholesterol in breast cancer. Endocrine-related cancer, 24(7), 339–349. https://doi.org/10.1530/ERC-16-0533
- Galvan, G. C., Friedrich, N. A., Das, S., Daniels, J. P., Pollan, S., Dambal, S., Suzuki, R., Sanders, S. E., You, S., Tanaka, H., Lee, Y. J., Yuan, W., de Bono, J. S., Vasilevskaya, I., Knudsen, K. E., Freeman, M. R., & Freedland, S. J. (2023). 27-hydroxycholesterol and DNA damage repair: implication in prostate cancer. Frontiers in oncology, 13, 1251297. https://doi.org/10.3389/fonc.2023.1251297
- Bilson, J., Scorletti, E., Swann, J. R., & Byrne, C. D. (2024). Bile Acids as Emerging Players at the Intersection of Steatotic Liver Disease and Cardiovascular Diseases. Biomolecules, 14(7), 841. https://doi.org/10.3390/biom14070841
- González-Ortiz, A., Galindo-Hernández, O., Hernández-Acevedo, G. N., Hurtado-Ureta, G., & García-González, V. (2021). Impact of cholesterol-pathways on breast cancer development, a metabolic landscape. Journal of Cancer, 12(14), 4307–4321. https://doi.org/10.7150/jca.54637


