Estrogen is one of the most physiologically significant steroid hormones in the human body, governing far more than reproductive function. Despite decades of intensive research, the complexity of estrogen signaling — involving multiple receptor subtypes, tissue-specific effects, and intricate metabolic pathways — continues to challenge both basic science and clinical translation. This article provides a comprehensive overview of estrogen biology, from its structural characteristics and biosynthetic origins to receptor signaling mechanisms, physiological and pathological roles, and the emerging role of multi-omics approaches in unraveling estrogen-related disease mechanisms.
1. What Is Estrogen? Types, Structures, and Classification
Estrogens are steroid hormones derived from cholesterol through the broader steroidogenic pathway. Their shared structural signature is an aromatic phenolic A ring, a defining feature that supports binding to estrogen receptors. Early estrogen research began in the 1920s, when ovarian extracts were shown to contain estrogenic activity; later purification and structural characterization established estrogens as a distinct class of steroid hormones.
1.1 Major Endogenous Estrogens: E1, E2, E3, and E4
The most commonly discussed endogenous estrogens in humans are estrone (E1), 17 beta-estradiol (E2), estriol (E3), and estetrol (E4). Although they share an 18-carbon steroid backbone, small differences in hydroxylation and oxidation patterns produce meaningful differences in receptor affinity, tissue activity, and physiological context (Chen et al., 2022).
| Estrogen | Structural Features | Potency / Receptor Affinity | Main Source / Context | Biological Relevance |
|---|---|---|---|---|
| Estradiol (E2) | Contains a phenolic hydroxyl group at C3 and a hydroxyl group at C17β. | The most potent natural estrogen; high binding affinity for estrogen receptors | Dominant circulating estrogen in premenopausal women | Central to ovarian function, reproductive cycling, bone maintenance, vascular biology, and many estrogen-responsive tissues |
| Estrone (E1) | Contains a phenolic hydroxyl group at C3 and a ketone group at C17. | Lower receptor affinity than E2 | Especially important after menopause; produced largely through aromatization of androstenedione in adipose tissue | Can be interconverted with E2 by 17β-hydroxysteroid dehydrogenase enzymes; contributes to postmenopausal estrogen activity |
| Estriol (E3) | Contains a phenolic hydroxyl group at C3, a hydroxyl group at C16α, and a hydroxyl group at C17β. | Lower estrogenic potency than E2 | Produced in high amounts during pregnancy through coordinated fetal-placental steroid metabolism | Reflects the activity of the fetal-placental unit and is biologically important during pregnancy |
| Estetrol (E4) | Contains a phenolic hydroxyl group at C3 and three additional hydroxyl groups at C15α, C16α, and C17β. | Lower overall affinity for estrogen receptors than E2, but with distinctive receptor pharmacology | Produced by the fetal liver during pregnancy | Has attracted attention because of its distinctive receptor pharmacology and clinical relevance in newer estrogen-containing therapeutic strategies |
1.2 Endogenous, Synthetic, and Environmental Estrogens
Estrogenic compounds can be classified by origin and function. Endogenous estrogens are produced within the body and include E1, E2, E3, and E4. Beyond endogenous estrogens, estrogenic compounds also include synthetic estrogens, estrogen receptor-targeting drugs, plant-derived phytoestrogens, and environmental xenoestrogens.
- Synthetic estrogens such as ethinyl estradiol are used in contraceptive and hormone therapy contexts.
- Selective estrogen receptor modulators (SERMs), selective estrogen receptor degraders (SERDs), and estrogen receptor antagonists are pharmacological agents designed to modulate, block, or degrade estrogen receptors in a tissue- or disease-specific manner.
- Phytoestrogens, including genistein and daidzein, are plant-derived compounds that can interact with estrogen receptors with variable and generally weaker potency than estradiol.
- Xenoestrogens, such as bisphenol A and certain endocrine-disrupting chemicals, are exogenous compounds that may interfere with normal estrogen signaling and are therefore important in toxicology, endocrinology, and environmental health research.
2. Estrogen Biosynthesis, Metabolism, and Receptor Signaling
Estrogen activity is controlled by three connected layers: biosynthesis, metabolic transformation, and receptor signaling. A change in any layer can alter downstream physiology or disease risk.
2.1 Estrogen Biosynthesis
Estrogen biosynthesis begins with cholesterol and proceeds through steroidogenic intermediates. Pregnenolone, progesterone, androstenedione, and testosterone serve as upstream precursors in the steroid hormone network. The terminal estrogen-defining reaction is aromatization, catalyzed by aromatase (CYP19A1), which converts androstenedione to estrone and testosterone to estradiol (Xu et al., 2022).
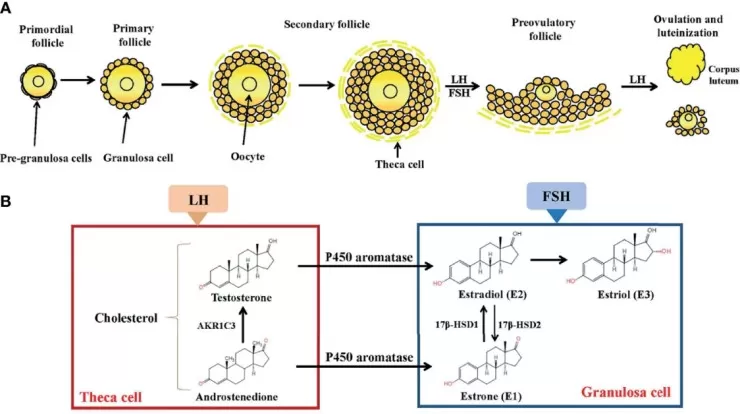
Figure 1. Folliculogenesis and follicular estrogen synthesis. Image reproduced from Xu et al., 2022, Frontiers in Endocrinology, 13, 827032.
Ovarian granulosa cells are the primary source of circulating estrogens in premenopausal women. Theca cells provide androgen precursors through CYP17A1 activity, and granulosa cell aromatase converts these androgens into estrogens. In postmenopausal women and in peripheral tissues, aromatase expression in adipose tissue, bone, skin, brain, and breast tissue becomes the dominant source of local estrogen production. This peripheral aromatization is especially relevant to obesity-associated breast cancer risk and metabolic disease (Chen et al., 2022).
2.2 Estrogen Metabolism
Estrogen metabolism regulates both hormone activity and hormone clearance. After synthesis, estrogens can be converted into multiple oxidative and conjugated metabolites, and these metabolic routes influence how long estrogens remain active, how they are transported, and how they are eliminated from the body.
Phase I metabolism mainly involves cytochrome P450-mediated hydroxylation. CYP1A1 and CYP1A2 are commonly associated with 2-hydroxylation, CYP1B1 contributes strongly to 4-hydroxylation, and CYP3A family enzymes can participate in 16α-hydroxylation and other oxidative reactions. These pathways generate metabolites such as 2-hydroxyestrone, 4-hydroxyestrone, and 16α-hydroxyestrone. Because different hydroxylation routes may produce metabolites with different biological properties, estrogen metabolite patterns have been studied in relation to oxidative stress, DNA damage, proliferative signaling, and hormone-dependent disease mechanisms (Almeida et al., 2021; Al-Shami et al., 2023).
Phase II metabolism generally promotes detoxification and excretion. Catechol-O-methyltransferase (COMT) methylates catechol estrogens, while uridine diphosphate glucuronosyltransferases (UGTs) and sulfotransferases (SULTs) catalyze glucuronidation and sulfation. These reactions increase metabolite solubility and support renal or biliary elimination. Together, Phase I and Phase II metabolism shape the estrogen metabolic profile, which can vary by tissue type, menopausal status, genetic background, disease state, and environmental exposure.
2.3 Estrogen Receptors and Signaling Mechanisms
Estrogens act mainly through estrogen receptor alpha (ERα, encoded by ESR1), estrogen receptor beta (ERβ, encoded by ESR2), and G protein-coupled estrogen receptor 1 (GPER1). ERα and ERβ are nuclear receptor family members that can regulate gene expression through direct binding to estrogen response elements (EREs), tethering to transcription factors such as AP-1 and Sp1, and recruiting co-activator or co-repressor complexes. ERα is especially important in uterus, mammary gland, liver, bone, and hypothalamic regulation, while ERβ shows broader roles in ovary, prostate, immune cells, vascular tissue, and the central nervous system (Chen et al., 2022; Clusan et al., 2023).
Estrogen signaling is not limited to slow transcriptional responses. Membrane-associated ER pools and GPER1 can activate rapid non-genomic signaling cascades, including PI3K/AKT, MAPK/ERK, cAMP, calcium signaling, and nitric oxide production. These pathways can cross-talk with receptor tyrosine kinases and growth-factor networks (Gan et al., 2024). In ER-positive breast cancer, such cross-talk is a key reason why estrogen signaling, kinase pathway activation, and endocrine therapy resistance are often studied together (Clusan et al., 2023).
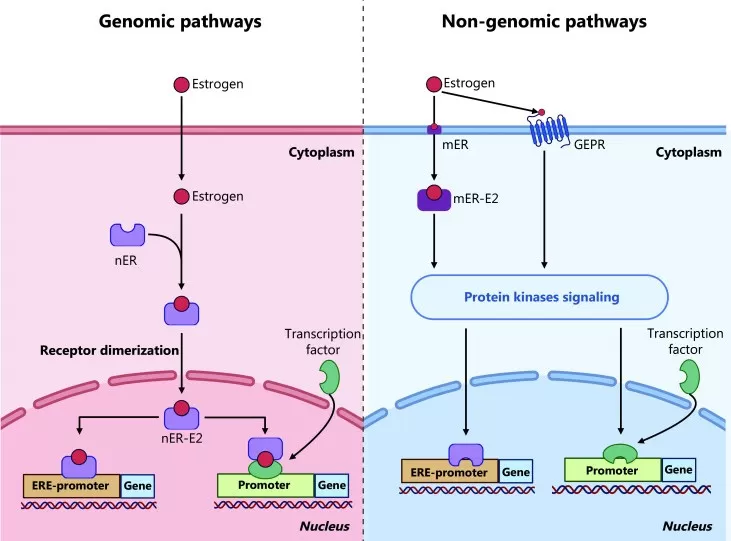
Figure 2. The genomic and non-genomic pathways of estrogen signaling. Image reproduced from Gan et al., 2024, Cancer Biology & Medicine, 21(10), 898–915.
3. Estrogen in Health and Disease: Physiological Roles and Pathological Links
Estrogen regulates far more than reproductive function. Through estrogen receptors and tissue-specific signaling networks, it contributes to reproductive cycling, bone remodeling, cardiovascular homeostasis, metabolic regulation, brain function, and immune balance. These effects are highly context-dependent: the same signaling pathways that support normal physiology may contribute to disease when estrogen production, metabolism, receptor expression, or downstream signaling becomes dysregulated. Understanding estrogen in both health and disease therefore requires a system-level view of hormone activity across tissues and life stages.
3.1 Estrogen in Reproductive Biology and Hormone-Dependent Cancers
In the female reproductive system, estradiol regulates follicular development, endometrial proliferation, cervical mucus changes, and hypothalamic-pituitary-gonadal axis feedback. During the follicular phase, rising E2 promotes endometrial growth and prepares the reproductive tract for potential implantation. Sustained high E2 levels near mid-cycle contribute to the luteinizing hormone surge and ovulation. After ovulation, estrogen works with progesterone to support endometrial receptivity. Estrogen also contributes to mammary gland development, ductal morphogenesis, and pregnancy-associated tissue remodeling. In males, estrogen supports spermatogenesis, bone homeostasis, libido, and metabolic regulation, showing that estrogen biology is not limited to female physiology (Chen et al., 2022).
When estrogen signaling becomes dysregulated, it can contribute to hormone-dependent disease. Breast cancer is the most extensively studied example. Approximately 70% of breast cancers are estrogen receptor-positive, and ERα signaling can promote tumor cell proliferation and survival. In advanced disease, endocrine resistance may involve ESR1 mutations, altered co-regulator activity, PI3K/AKT/mTOR pathway activation, cell-cycle remodeling, and tumor microenvironment effects (Clusan et al., 2023). Estrogen metabolism may also be relevant to breast carcinogenesis, as catechol estrogen formation, insufficient Phase II detoxification, estrogen quinone formation, oxidative stress, and DNA adduct formation have been investigated as potential mechanisms contributing to estrogen-associated breast cancer risk (Almeida et al., 2021; Al-Shami et al., 2023).
Endometrial cancer is also closely linked to estrogen exposure. Prolonged estrogen stimulation that is insufficiently opposed by progesterone can promote endometrial hyperplasia and increase the risk of malignant transformation. This mechanism is particularly relevant in anovulatory cycles, obesity, estrogen-only hormone therapy in individuals with an intact uterus, and other settings that increase endometrial estrogen exposure. Ovarian cancer has a more heterogeneous relationship with estrogen signaling. Estrogen receptors are expressed in some ovarian tumor subtypes, but the biological role of estrogen depends strongly on tumor histotype, receptor profile, and disease context.
3.2 Estrogen in Bone, Cardiometabolic Health, and Metabolic Disease
Estrogen is essential for maintaining skeletal homeostasis. It helps balance bone formation and bone resorption by suppressing excessive osteoclast activity, supporting osteoblast survival and function, and influencing the RANKL/osteoprotegerin axis and inflammatory cytokine activity. When estrogen levels decline after menopause, bone resorption accelerates, increasing the risk of low bone mass and postmenopausal osteoporosis (Chen et al., 2022; Wu et al., 2021).
Estrogen also contributes to cardiovascular health. It has been associated with endothelial nitric oxide production, lipid metabolism, vascular inflammation, and blood vessel flexibility. These effects may partly explain why premenopausal women generally have lower cardiovascular disease risk than age-matched men. However, cardiovascular disease is multifactorial, and estrogen is only one part of a broader risk landscape that includes age, blood pressure, obesity, diabetes, lifestyle, and genetic background (Chen et al., 2022; den Ruijter & Kararigas, 2022).
Metabolic changes are also common during the menopause transition. As estrogen levels decline, many women experience increased visceral fat, reduced insulin sensitivity, unfavorable lipid changes, and a higher risk of metabolic disorders. Metabolic dysfunction-associated steatotic liver disease (MASLD), formerly known as NAFLD, is one estrogen-relevant metabolic condition. After menopause, MASLD prevalence and severity tend to increase. Current evidence suggests that estrogen deficiency may contribute to liver fat accumulation, insulin resistance, inflammation, and disease progression, while obesity, diabetes, diet, physical activity, age, and genetic background remain major determinants of MASLD risk (Dong et al., 2025).
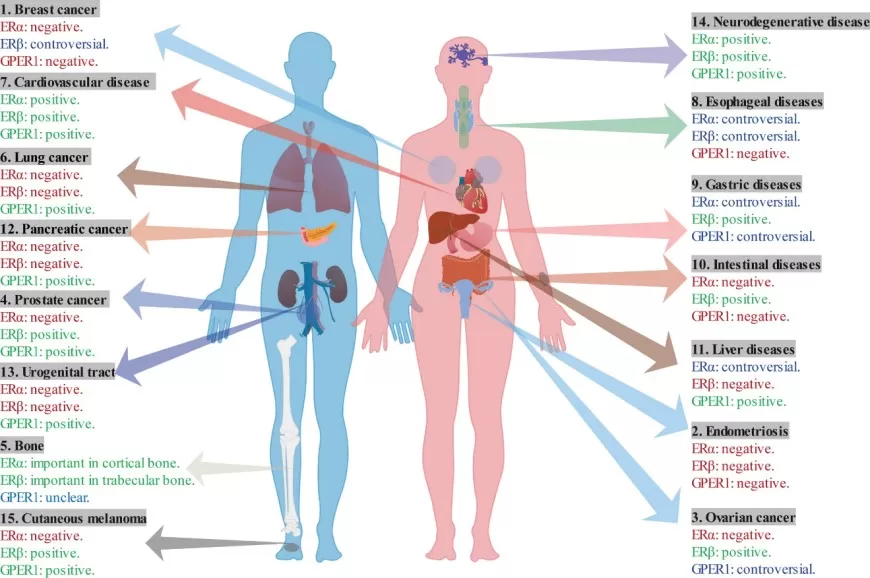
Figure 3. The multifaceted role of ERs in various diseases. Image reproduced from Chen et al., 2022, Frontiers in Endocrinology, 13, 839005.
3.3 Estrogen in Brain Function and Neurodegenerative Disease
Estrogen receptors are expressed in brain regions involved in cognition, mood, thermoregulation, and neuroendocrine control. Experimental studies suggest that estrogen can influence synaptic plasticity, mitochondrial function, cholinergic signaling, oxidative stress, and neuroinflammation. These functions make estrogen relevant to studies of menopausal symptoms, brain aging, and neurodegenerative disease.
In Alzheimer's disease research, one important concept is the “timing hypothesis.” This hypothesis proposes that menopausal hormone therapy may have different effects depending on whether it is initiated near the menopausal transition or many years later. However, clinical evidence remains mixed, and hormone therapy should not be interpreted as a simple preventive strategy for dementia (Nerattini et al., 2023).
3.4 Estrogen in Immune Regulation and Autoimmune Conditions
Estrogen is also involved in immune regulation. It can influence B cell survival, T helper cell polarization, cytokine production, antigen presentation, and inflammatory signaling. These effects may contribute to sex-biased patterns in immune responses and autoimmune disease activity (Cutolo & Straub, 2020).
Systemic lupus erythematosus, rheumatoid arthritis, and multiple sclerosis are examples of autoimmune diseases with notable sex differences in prevalence or disease behavior. Estrogen may contribute to immune activation or immune modulation depending on concentration, receptor subtype, immune cell type, and disease stage. However, autoimmune disease risk is not determined by estrogen alone. Sex chromosomes, pregnancy-related immune adaptation, microbiome factors, environmental exposures, and genetic susceptibility also shape autoimmune disease onset, progression, and severity.
4. Therapeutic Mechanisms in Estrogen-Related Diseases
Estrogen-related diseases, particularly hormone-dependent cancers, have driven sustained interest in therapies that modulate estrogen production, receptor activity, or downstream signaling. From a mechanistic perspective, current therapeutic strategies generally fall into several categories. Some approaches reduce estrogen biosynthesis by inhibiting aromatase-dependent conversion of androgens to estrogens, thereby lowering ligand availability. Others interfere directly with estrogen receptor signaling by blocking receptor activation, altering tissue-specific receptor responses, or promoting receptor degradation. Additional strategies target downstream pathways that cooperate with estrogen receptor activity, including cell-cycle regulation, growth factor signaling, and kinase-mediated resistance mechanisms (Clusan et al., 2023).
These therapeutic principles highlight the complexity of estrogen biology. Disease progression and therapeutic resistance may involve altered estrogen metabolism, receptor mutations or remodeling, co-regulator imbalance, pathway cross-talk, and metabolic adaptation. Therefore, estrogen-related research increasingly requires more than single-molecule measurement or single-pathway analysis. Mechanism-oriented studies benefit from integrated analytical strategies that can capture hormone levels, estrogen metabolites, receptor-associated proteins, phosphorylation events, and broader pathway remodeling. This need creates a natural transition from therapeutic concepts to omics-based research methods.
5. Omics Research Methods in Estrogen Studies
Estrogen research increasingly requires analytical strategies that can quantify hormone molecules, capture pathway activity, and connect molecular changes with disease phenotypes. For this topic, metabolomics and proteomics are especially central because they directly measure steroid hormone metabolism, protein abundance, post-translational modifications, and receptor pathway effects.
5.1 Metabolomics for Estrogen Metabolism and Pathway Discovery
Metabolomics is well suited for estrogen studies because it can quantify parent hormones, hydroxylated and methoxylated metabolites, conjugated forms, and downstream metabolic shifts. Targeted LC-MS/MS is preferred when predefined steroid hormones or estrogen metabolites require sensitive quantification, while untargeted metabolomics can reveal broader pathway changes related to lipid, amino acid, or energy metabolism. In a nested case-control study, Falk et al. measured 15 serum estrogens and estrogen metabolites by LC-MS/MS in postmenopausal women and showed that pathway-level estrogen metabolite patterns may provide information beyond parent estrogen levels (Falk et al., 2013).
5.2 Proteomics and Modification Proteomics for Estrogen Signaling
Proteomics helps characterize estrogen-responsive proteins, receptor-associated networks, and pathway-level remodeling that cannot be fully inferred from transcript abundance. Modification proteomics, especially phosphoproteomics, is valuable for studying rapid estrogen signaling, kinase activation, and pathway cross-talk. Cuesta et al. applied phosphoproteome analysis to ER-positive MCF7 breast cancer cells treated with estrogen and identified estrogen-regulated mTORC1 targets, including DEPTOR. This study showed how phosphoproteomics can connect estrogen receptor activity with PI3K/AKT/mTOR signaling and reveal regulatory mechanisms relevant to endocrine-response biology (Cuesta et al., 2019).
5.3 Multi-Omics Integration for Mechanism-Level Interpretation
Multi-omics integration links molecular layers such as gene expression, metabolite abundance, protein regulation, and pathway activity, making it useful when estrogen effects involve coordinated transcriptional and metabolic remodeling. This approach can identify regulatory networks that single-omics analysis may miss. Shen et al. integrated transcriptomics and metabolomics in estradiol-treated HepG2 liver cancer cells and found that estrogen and estrogen receptor agonists reshaped gene-metabolite interactions related to glycolysis, oxidative phosphorylation, amino acid metabolism, and cell growth. The study illustrates how integrated omics can connect estrogen receptor signaling with functional metabolic phenotypes (Shen et al., 2021).
How MetwareBio Supports Estrogen-Related Research
Estrogen-related research requires precise measurement of steroid hormones and system-level characterization of molecular response. MetwareBio is a leading multi-omics CRO providing LC-MS-based metabolomics, mass spectrometry-based proteomics, and integrated multi-omics analysis services for biomedical, pharmaceutical, agricultural, and clinical research. For estrogen-related studies, MetwareBio's targeted steroid hormone metabolomics service offers a practical solution for quantifying estrogen-related hormones and pathway-associated steroid molecules in suitable biological matrices.
This service can support studies of hormone-dependent cancer, reproductive biology, menopause-related metabolic changes, endocrine disruption, pregnancy-associated steroid remodeling, MASLD-related hormone changes, and hormone-treatment response. When broader mechanism interpretation is required, targeted steroid hormone profiling can be combined with MetwareBio's untargeted metabolomics, proteomics, phosphoproteomics, lipidomics, and multi-omics integration services to connect hormone-level changes with metabolic pathways, receptor-associated signaling, and biomarker discovery.
For each service project, MetwareBio provides end-to-end support from experimental design and sample preparation to data analysis, pathway interpretation, and result visualization. For more information, please contact MetwareBio to discuss a suitable omics strategy for an estrogen-related research project.
Contact UsRead More: Steroid Hormones, Signaling, and Omics Approaches
Explore related articles on cholesterol metabolism, oxysterol biology, estrogen receptor signaling in cancer, and multi-omics integration strategies for hormone-related research.
Cholesterol is the upstream precursor for all steroid hormones including estrogen. This article covers cholesterol biosynthesis, transport, homeostatic regulation, and how metabolomics and proteomics are applied to study cholesterol-related metabolic diseases.
27-HC is an oxysterol with estrogen receptor modulating activity that links cholesterol metabolism to hormone-dependent cancer progression. This article details the dual role of 27-HC as both an ER agonist and an LXR ligand in tumor biology.
As another key oxysterol downstream of cholesterol, 25-HC bridges lipid metabolism with immune regulation and inflammatory signaling. This article examines how oxysterol measurement connects with estrogen-related immune modulation research.
This article provides a systematic overview of the complete steroidogenic pathway from cholesterol to estrogens, androgens, progestogens, glucocorticoids, and mineralocorticoids — a foundational reference for understanding estrogen biosynthesis in context.
References
- Almeida, M., Soares, M., Fonseca-Moutinho, J., Ramalhinho, A. C., & Breitenfeld, L. (2021). Influence of estrogenic metabolic pathway genes polymorphisms on postmenopausal breast cancer risk. Pharmaceuticals, 14(2), 94. https://doi.org/10.3390/ph14020094
- Al-Shami, K., Awadi, S., Khamees, A., Alsheikh, A. M., Al-Sharif, S., AlaBereshy, R., Al-Eitan, S. F., Banikhaled, S. H., Al-Qudimat, A. R., Al-Zoubi, R. M., & Al Zoubi, M. S. (2023). Estrogens and the risk of breast cancer: A narrative review of literature. Heliyon, 9(9), e20224. https://doi.org/10.1016/j.heliyon.2023.e20224
- Chen, P., Li, B., & Ou-Yang, L. (2022). Role of estrogen receptors in health and disease. Frontiers in Endocrinology, 13, 839005. https://doi.org/10.3389/fendo.2022.839005
- Clusan, L., Ferrière, F., Flouriot, G., & Pakdel, F. (2023). A basic review on estrogen receptor signaling pathways in breast cancer. International Journal of Molecular Sciences, 24(7), 6834. https://doi.org/10.3390/ijms24076834
- Cuesta, R., Gritsenko, M. A., Petyuk, V. A., Shukla, A. K., Tsai, C.-F., Liu, T., McDermott, J. E., & Holz, M. K. (2019). Phosphoproteome analysis reveals estrogen-ER pathway as a modulator of mTOR activity via DEPTOR. Molecular & Cellular Proteomics, 18(8), 1607–1618. https://doi.org/10.1074/mcp.RA119.001506
- Cutolo, M., & Straub, R. H. (2020). Sex steroids and autoimmune rheumatic diseases: State of the art. Nature Reviews Rheumatology, 16, 628–644. https://doi.org/10.1038/s41584-020-0503-4
- den Ruijter, H. M., & Kararigas, G. (2022). Estrogen and cardiovascular health. Frontiers in Cardiovascular Medicine, 9, 886592. https://doi.org/10.3389/fcvm.2022.886592
- Dong, J., Dennis, K. M. J. H., Venkatakrishnan, R., Hodson, L., & Tomlinson, J. W. (2025). The impact of estrogen deficiency on liver metabolism: Implications for hormone replacement therapy. Endocrine Reviews, 46(6), 790–809. https://doi.org/10.1210/endrev/bnaf018
- Falk, R. T., Brinton, L. A., Dorgan, J. F., Fuhrman, B. J., Veenstra, T. D., Xu, X., & Gierach, G. L. (2013). Relationship of serum estrogens and estrogen metabolites to postmenopausal breast cancer risk: A nested case-control study. Breast Cancer Research, 15, R34. https://doi.org/10.1186/bcr3416
- Gan, X., Dai, G., Li, Y., Xu, L., & Liu, G. (2024). Intricate roles of estrogen and estrogen receptors in digestive system cancers: a systematic review. Cancer Biology & Medicine, 21(10), 898–915. https://doi.org/10.20892/j.issn.2095-3941.2024.0224
- Nerattini, M., Jett, S., Andy, C., Carlton, C., Zarate, C., Boneu, C., Battista, M., Pahlajani, S., Loeb-Zeitlin, S., Havryulik, Y., Williams, S., Christos, P., Fink, M., Brinton, R. D., & Mosconi, L. (2023). Systematic review and meta-analysis of the effects of menopause hormone therapy on risk of Alzheimer's disease and dementia. Frontiers in Aging Neuroscience, 15, 1260427. https://doi.org/10.3389/fnagi.2023.1260427
- Shen, M., Xu, M., Zhong, F., Crist, M. C., Prior, A. B., Yang, K., Allaire, D. M., Choueiry, F., Zhu, J., & Shi, H. (2021). A multi-omics study revealing the metabolic effects of estrogen in liver cancer cells HepG2. Cells, 10(2), 455. https://doi.org/10.3390/cells10020455
- Wu, D., Cline-Smith, A., Shashkova, E., Perla, A., Katyal, A., & Aurora, R. (2021). T-cell mediated inflammation in postmenopausal osteoporosis. Frontiers in Immunology, 12, 687551. https://doi.org/10.3389/fimmu.2021.687551
- Xu, X. L., Huang, Z. Y., Yu, K., Li, J., Fu, X. W., & Deng, S. L. (2022). Estrogen Biosynthesis and Signal Transduction in Ovarian Disease. Frontiers in Endocrinology, 13, 827032. https://doi.org/10.3389/fendo.2022.827032


